Mauveine: History, Properties and Structure
| ✅ Paper Type: Free Essay | ✅ Subject: Chemistry |
| ✅ Wordcount: 1267 words | ✅ Published: 30 Jan 2018 |
- Introduction
One of the most important events of 1856 was the discovery of the first successful artificial dye, mauveine (aniline purple, mauve dye, Tyrian purple) by William Perkin. The discovery of mauveine led to rapidly growth of fashion trend and discovery of new synthetic dyes. Nowadays there are more than three million dyes (WordPress.com). Most dyes used back in the XIX century were derived from natural products such as animals, vegetables or minerals. The dyes made from natural products at that time were expensive and required a large amount of labour. The main disadvantage of natural dyes was their tendency to fade.
The invention of mauveine was major landmark in the history of technology and science which brought great benefits to textile and synthetic dye industry. However most people do not aware of the origin of those colours, especially world’s first synthetic dye mauveine and chemical reactions beyond the mauveine dye production. This paper will discuss the historical background of mauveine invention, chemical structure of mauveine, chemical synthesis methods of mauveine and applications.
- Historical background
In 1834, German chemist August Wilhem Hofmann was working derivatives of coal tar to produce a quinine substance for treatment of malaria. At that time, young William Perkin was one of the Hofmann’s assistants and embarked to solve this problem. In 1856, while Hofmann went back to visit Germany, Perkin conducted some experiments in the laboratory at his home to achieve Hofmann’s goal. Perkin oxidised the aniline in the presence of potassium dichromate and obtained black coloured sludge. At the end of the experiment Perkin diluted the black sludge with alcohol and gained a purple solution. Perkin patented his new discovery and established the company ‘Perkin and Sons’. In June 1857 the company ‘Perkin and Sons’ began its work producing synthetic dyes.
It should be emphasized that many technical problems occurred during the operation of the company such as large scale of benzene nitration and nitrobenzene reduction. The aniline purple was obtained in poor yields, because the oxidation step required optimisation.
By 1864, the company was able to trade its product as a crystalline solid. In spite of the wealth which came with successful dye production, Perkin continued his experimental works in chemistry. After his successful introduction of aniline purple in Britain, next step for Perkin was to export mauveine abroad. However, Perkin’s effort to introduce his invention in France ended unsuccessfully as his patent was invalidated. Invention of the Perkins aniline purpleencouraged other chemists to work on new synthetic dyes. For instance, in 1856 Natanson discovered magenta (triphenylmethane dye) by oxidising aniline with chloride. As it was mentioned above, developments of synthetic dye chemistry in modern times takes its background from mauveine. Taking the historical background into consideration, mauveine made our world more colorful than it was before(‘the chemistry and application of dyes’, edited by David R. Waring, and Geoffrey Hallas, Plenum Press, New York 1990, page 4-5).
- Chemical structure and properties of mauveine.
The structure and consist of mauveine for many years had remained mystery. First attempts establishing mauveine molecular structure was made by Perkin himself. Perkin derived similar dye when oxidised toluidine and xylidine. However, pure aniline gave dye with poorer quality than that obtained from original impure aniline. This product was called pseudomauveine which was derivative of mauveine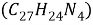 . Perkin concluded that mauveine contains both pseudo-mauveine (
. Perkin concluded that mauveine contains both pseudo-mauveine ( ) and a trimethyl derivative. At that time Perkin could not determine every atom in the molecule and so he attempted to determine mauveines constitutional formula by identified properties of mauve. In 1879 Perkin came to conclusion that mauveine can be derived only from aniline and para-toluidine. It should be emphasized that Perkins work determined a molecular formula of mauveine but not a molecular structure. Other works to determine molecular structure of mauveine was done by Fischer, Hepp and Nietzki. By synthesising pseudo-mauveine in several times they discovered pseudomauveines structure. Oxidative couplings were involved in this synthesis as shown in figure 1.
) and a trimethyl derivative. At that time Perkin could not determine every atom in the molecule and so he attempted to determine mauveines constitutional formula by identified properties of mauve. In 1879 Perkin came to conclusion that mauveine can be derived only from aniline and para-toluidine. It should be emphasized that Perkins work determined a molecular formula of mauveine but not a molecular structure. Other works to determine molecular structure of mauveine was done by Fischer, Hepp and Nietzki. By synthesising pseudo-mauveine in several times they discovered pseudomauveines structure. Oxidative couplings were involved in this synthesis as shown in figure 1.
 Synthetic methods for pseudomauveine figure 1.
Synthetic methods for pseudomauveine figure 1.
However, there was little evidence to proof mauveines chemical structure (figure 2).

Mauveine figure 2.
Only in 1994, the structure of mauveine determined through the work of Meth-Cohn and Smith. They emphasised two compounds as a key chromophores to obtain mauveine, mauveine A with C26 structure and mauveine B with C27 structure (scheme 1). Meth-Cohn and Smith analysed seven dyed textiles and seven mauve salts from different museum such as Science museum in London (scML), Chandler Museum (Columbia University), Museum of Science and Industry in Manchester (MSIM) and Perth Museum in Scotland. Meth-Cohn and Smith analysed major components of those samples by mass spectroscopy and NMR spectrometry and found presence of different methylated homologues of pseudo-mauveine. Different chromospheres are shown in table 1 amongst the historical samples which Meth-Cohn and Smith had found.
|
|
|
A |
B |
B2 |
B3+B4 |
C |
C1 |
|
|
|
ScML1 |
1 |
2 |
50 |
23 |
10 |
5 |
4 |
5 |
1.3 |
|
ScML2 |
1 |
3 |
37 |
26 |
13 |
6 |
5 |
8 |
0.8 |
|
ScML3 |
1 |
2 |
54 |
16 |
9 |
4 |
5 |
8 |
1.8 |
|
ScML4 |
1 |
2 |
37 |
31 |
12 |
5 |
5 |
8 |
0.8 |
|
MSIM1 |
– |
2 |
39 |
33 |
12 |
5 |
4 |
6 |
0.8 |
|
MSIM2 |
49 |
41 |
7 |
– |
3 |
– |
– |
– |
– |
|
CM |
1 |
2 |
50 |
24 |
8 |
4 |
5 |
7 |
1.4 |
Table1. Percentage of chronophers which found from mauveine samples
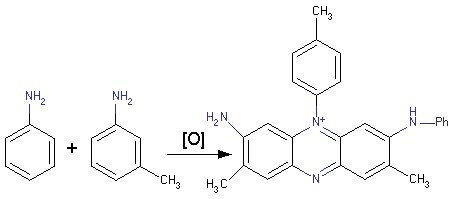
Meth-Cohn and Smith discovered that all mauveine samples which prepared by Perkin was mixture of about thirteen different compounds with 7-amino-5-phenyl-3-(phenylamino)-phenazin-5-ium core. The majority of the historical samples of mauve which were investigated by Meth-Cohn and Smith had both A or B mauveines. Apart from that almost all historical samples of mauveine contained mono, tetra methylated derivatives of pseudo-mauveine. Mauveine B2 (C27) and C25A, C25B were important fingerprint markers of textiles and salts amongst these. In addition, for the first time Meth-Cohn and Smith identified pseudo-mauveine in the historical sample, this was described by Perkin as a second colouring source.
As it was mentioned above, the mauveine contained pseudo-mauveine, which is very soluble, uncrystallisable salt and another one is less soluble, crystalline salt 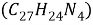 obtained from para-toluidine and aniline. One of the important properties of mauveine is fastness with silk which they colour.
obtained from para-toluidine and aniline. One of the important properties of mauveine is fastness with silk which they colour.
- Chemical synthesis methods of mauveine
As mentioned in previous sections William Perkin did not discover the mauveine intentionally, his main aim was preparing quinine ( ). As chemistry were in the early stage of its development there were little known about quinines structural formula. Taking into account of the molecular formulas of quinine and allyl toluidine Perkin came to the relationship shown in equation 1 and he attempted the preparation of quinine by oxidising allyl toluidine in the presence of potassium dichromate in sulphuric acid:
). As chemistry were in the early stage of its development there were little known about quinines structural formula. Taking into account of the molecular formulas of quinine and allyl toluidine Perkin came to the relationship shown in equation 1 and he attempted the preparation of quinine by oxidising allyl toluidine in the presence of potassium dichromate in sulphuric acid:
|
|
Equation1 |
In his note Perkin wrote ‘that no quinine was formed, but only a dirty reddish brown precipitate’.
However, Perkin was not aware at that time structure of allyl toluidine and quinine. If he had known the structure, he would certainly choose other route for synthesising quinine:
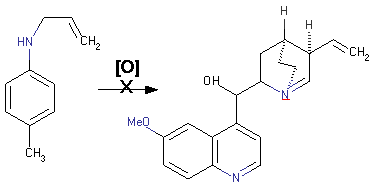
The synthesis of mauveine starting aniline also required toluidine impurities (equation 2):
- Application of mauveine
Alcoholic extract of mauveine gives very brilliant purple colour which cannot be removed and stable during washing. Perkin quickly considered its potential as a dyestuff for textiles and fabrics.
Another useful application of mauveine is food colouring dye. In United States during 1900th the mauveine was used as food colouring with different names such as violate paste, rosolan, Perkin’s violate.
- Conclusion
This short essay has considered history of the mauveine discovery, chemical structure and properties, synthesising way of mauveine. The discovery of mauveine led to rapidly change in global fashion and chemical industry. The Perkins discovery cannot be judged by today’s knowledge and consider as a luck. There were many things which have been discovered and invented, however they were forgotten as it did not find any practical application. Perkin had the ability to combine the theoretical and practical work from his experiment. Perkin’s discovery still has not lost its significance in modern synthetic dye chemistry and contributed to development of modern fashion.
Galymzhan


Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allDMCA / Removal Request
If you are the original writer of this essay and no longer wish to have your work published on UKEssays.com then please click the following link to email our support team:
Request essay removal










