Impact of Bisphenol A in Bottles on Children’s health
| ✅ Paper Type: Free Essay | ✅ Subject: Chemistry |
| ✅ Wordcount: 2412 words | ✅ Published: 26 Jan 2018 |
|
Name of student: Le Tien Dat |
- INTRODUCTION
The history of Bisphenol-A is a long story, and the development of this substance has gathered benefits and drawbacks. In fact, BPA brings several benefits for life’s people. It is used in the production of epoxy resins and polycarbonate plastics. The polycarbonate plastics may be encountered in many products including dental equipment, eyewear, especially baby bottles, while epoxy resins are frequently used as inner liner of metal food and drink containers to prevent corrosion.
However, historical development of Bisphenol A demonstrates that the drawbacks have a much negative impact than benefits. Bisphenol A was discovered in 1891 by Russian chemist Aleksandr Dianin. In the 1930s, scientists discovered that BPA is an artificial estrogen and it has been used widely over the world since 1950 (The Washington Post, “117 Years of BPA,” April 27, 2008). Recently, the number and diversity in the study of the effects of bisphenol A has grown significantly over the past few years. In 2009, Health Canada studies showed that the majority of canned soft drinks contain bisphenol A (Survey of Bisphenol A in Canned Drink Products,13 March2009). Moreover, in 2010, the University of Texas in USA showed that almost plastic packaging and canned milk usually contain high the percentage of BPA[Journalist’s Resource.org]. In addition, study in the US showed that consumption of soda, school lunches, and meals prepared outside the home make a significant contribution to the increasing BPA in the urine [Lakind JS, Naiman DQ (2010)]. Therefore, the wide use of BPA in plastic fabrication and epoxy resins has a negative impact on health people, which interferes with the endocrine system in animals and humans and contribute to adversely affect children’s health. Furthermore, plastic containers, especially baby bottles, become more popular with consumers and retailers as they are cheap, functional and hygienic way to transport products. This leads to the fact that many children will be easily caught disease as well as influence the development of children.
This essay will discuss about concentrations of BPA in urine which can cause danger for children ages from 6 to 17 as well as the impact of Bisphenol A in bottles on children, such as early puberty, obesity and lower IQ (Csaba Leranth, 2013), and the positive solutions to minimize problems such as a ban, use plastic bottles as less as possible.
- SITUATION
The discovery of Bisphenol A in the 20 century was just an accident. Although Aleksandr Dianin, Russian chemist, discovered Biisphenol A in 1891, until the year 1900, Dr. Feldman made some important discoveries of this. In 1990, Dr. Feldman and his colleagues have carried out a research into estrogen, and they found out strange substance that looked like an estrogen. In 1992, strange substance was found in experimentation related to plastic flask and was not present in glass flask. Dr. Feldman identified this substance as Bisphenol A. According to International Union of Pure and Applied Chemistry Nomenclature (IUPAC), Bisphenol A (BPA) or 4,4′-dihydroxy-2,2-diphenylpropane is a carbon-based synthetic compound with the chemical formula C15H16O2 and molecular weight 228.28634 g/mol (Bisphenol A, 2005). This substance was used in the manufacture of polycarbonate plastic, which typically present in bottles. Furthermore Dr. Feldman said that Bisphenol A is a major health risk.
Nevertheless, the negative effect of Bisphenol A that Dr. Feldman research do not help inhibit the increasing manufacture of bottles, which contain high-level BPA, and the provided pie chart below illustrates the figure for baby bottles purchase worldwide.

Sources: Icon Group “The 2009-2014 World Outlook for Baby Bottles”- Philip M. Parker, PhD/SGD
At first glance, the manufacture of bottles in North America experienced the highest portion while Indian gained the least attention in this field. It is noticeable that 24 was the percentage that mass-produce bottles in North America, compared to 22% of Europe and 14% of Russia, Africa and Middle East. However India and Latin America just put the modest manufacture of bottles, which was 5% and 8% respectively. In 2008, there were 22 billion food cans and 100 billion cans for soft drinks, which were produced with BPA. (Hemi Weingarten, 2009).
With the innumerable bottles produced each year, people, and children in particularly, are easily expose to Bisphenol a contained in bottles. The number of children whose exposure to BPA had increased significantly since BPA was used widely from 1950. Furthermore, Karin B. Michels, associate professor of epidemiology at HSPH and Harvard Medical School said that drinking water from polycarbonate bottles in just one week went up urinary BPA levels more than two third, and if people heat those bottles, the levels would be higher (Karin B, 2015).
The line graph below demonstrates the concentrations in urine samples in children.
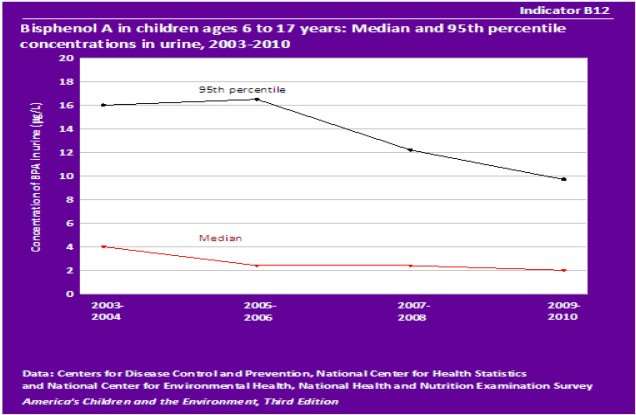 (Source: http://www.epa.gov/ace/biomonitoring/bpa.html)
(Source: http://www.epa.gov/ace/biomonitoring/bpa.html)
To find out whether or not children expose to Bisphenol A, many scientists research in urine samples from all age groups, including children and adults. This data which show concentrations in urine samples are obtained from individual survey participants conducted by National Center for Health Statistics and National Center for Environmental Health, National Health and Nutrition Examination Survey. The figure for the Median and 95th percentile concentrations in urine decreased significantly. Between 2003 and 2004, the number of the median concentration of BPA in urine of children ages 6 to 17 years is 4 µg/L in comparison with 16 µg/L of 95th percentile concentration in urine. It is noticeable that the figure for 95th percentile concentrations in urine reached a peak at 17 µg/L in 2005-2006 while 2009 was the year that the Median experience the lowest percentage, about 2 µg/L. These changes are extremely important in reducing the rate children whose exposure to Bisphenol A.
- PROBLEMS
Although the current trend in using Bisphebol A had declined significantly, BPA pose risk to human health. Bishenol A was researched in several scientific reports of the independent research group over the world. According to a scenario analysis of 12 experts in the National Toxicology Program – NTP, BPA makes the reproductive system and the brain of newborn animals develop abnormally. A small dose of BPA can cause negative effects on young children. Some studies even suggest that young children are easily affected by Bisphenol A because the young body is still developing. Therefore, the immune system cannot protect and remove the dangerous chemical, especially Bisphenol A.
Generally, although eating too much or lack of exercise are the important factors that lead to obese in teenager, said Dr. De-Kun Li, Bisphenol A might also contribute to the increasing in the obesity rate. Li and his colleagues studied 1326 children aging from 9 to 12, and measured BPA levels in their urine. (Corvese, 2013). The girls who have higher BPA levels were associated with the double risk of obesity. Researchers explained that Bisphenol A is referred to as endocrine disrupting chemicals, which interfere with elimination of natural hormones in the body. These natural hormones are responsible for reproductive development, fertility and maintenance of homeostasis. Therefore, when the concentration of BPA are high in girl, Bisphenol A which acts like estrogen make a significant contribution to the early puberty and cause weight gain.
With the weakened immune system and the consequences of obesity caused by Bisphenol A, young people are more likely suffer diseases such as: high cholesterol, high blood pressure and diabetes (Ogden CL, Carroll MD, Kit BK, Flegal KM, 2011). People who become obese as early as age are more likely suffer cancer in adults including cancer of the breast, colon, kidney, pancreas, gall bladder, and prostate. (A Cancer Journal for Clinicians2006). In 2010, a report from The President’s cancer Panel identified more than 130 studies that have linked BPA to obesity, breast cancer and other disorder problems (World-wire.com, 2010). Besides, there are many serious consequences of BPA that are proved by many scientists, such as: brain and behavior problems, heart problems and low IQ (Empen, 2013). If these diseases are not treated effectively, they have a negative impact on the development of children in the future. Therefore, eliminating Bisphenol A from the body can help young children have an excellent health without affecting by prevalence diseases in modern life
- SOLUTIONS
Recognizing the harmful effects of Bisphenol A and considering it as a national issue, scientists and governments take this problem in serious concern.
From a scientific perspective, developing alternative to Bisphenol A is a major breakthrough in eliminating BPA from the body as well as in cancer research. Furthermore, under the pressure of people who worry about the BPA’s adverse affect on human health, researchers over the world put more effort in finding the substitute for Bisphenol A. In fact, many manufactures in United States show that it is possible to replace BPA not only in baby bottles, but also in food cans and drinking bottles. They developed a chemical which originate in sugar, and this substance allow us to create a family of isosorbide-based epoxy resins that is capable of standing in for Bisphenol A, said Professor Michael Jaffe (Harrington, 2010). Besides, there are many possible substitutes to bottles and containers containing BPA, such as stainless steel, Tritan Copolyester, Polypropylene (PP), Polyethylene Terephthalate (PET), Polyamide , Polyethersulfone (PES) and oleoresin [Oregon Environmental Council, 2013; Det Økologiske Råd, 2013], all of which are environmentally friendly alternatives. In 2008, Nalgene said that they produce baby bottles with these substitutes, and it is obvious that these materials do not contain Bisphenol A.
Apart from practical concerns mentioned above, it would be unfair without considering the contributing factor of government in wiping off Bisphenol A from baby bottles. Canada becomes the first government in the world to carry out the negative effects of Bisphenol A. In October 2010, they imposed limitations on using bottles which contain BPA. Apparently, these ban helped decrease the rate of using bottles. Not only does Canada’s government ban using Bisphenol A, but also other countries legislate on the reduction of baby bottles. Washington is a good illustration for campaigning to act against BPA. “Bans BPA from children’s food and beverage containers (other than metal cans) and all reusable water bottles.(Adopted in 2010)” according to Washington State Legislature. Besides, governments force manufactures to label each baby bottles with the contents and statistics of Bisphenol A. Hence government can manage the concentration of BPA in bottles. In addition, thanks to attaching label, individual can easily check the bottles whether or not contain BPA. If the products say “phthalate free” or “BPA-free”, consumers can purchase these products without worrying the harmful effect of Bisphenol A (Niehs.nih.gov, 2015).
- EVALUATION
As we know, nothing interesting is ever completely one-sided, these solutions that wipe Bisphenol A of baby bottles also have the pros and cons. It seems to me that the potential consequences of rules and regulations are significant than the possible dangers.
On the one hand, imposing constraints have a negative effect on the quality of products. Bisphenol A is a suitable chemical for hardening polycarbonate plastics. If government ban using Bisphenol A in baby bottles, the quality of these bottles will degrade. Furthermore, the substitute is not as good as Bisphenol A. For instance, acrylics, which stand in for Bisphenol A, are used in producing bottles. However, this substance is more brittle than Bisphenol A, and it is obvious that the bottles are easily broken.
On the other hand, I would argue that these drawbacks are outweighed by the benefits. Removing Bisphenol A will be of benefit to economy. In 2008, there were 12,404 cases of childhood obesity and 33,863 cases of coronary heart diseases that associated with Bisphenol A. By eliminating this substance from the body, we can reduce child health care expenditure, which was about $2.98billion. Specifically, the cost of BPA- attributable childhood obesity and coronary heart were approximately 1.5 billion and 935 million respectively (Trasande, 2014). . Furthermore, if we use the substitute for Bisphenol A, it costs a fortune. Some report suggest that oleoresin, which stand in for Bisphenol A, can cost 2.2 cent more than BPA in the lining of aluminum cans. With 100 billion these cans produced each year, the cost of substitute for BPA would be 2.2 billions [LaytonL, 2010].
- CONCLUSION
The development of Bisphenol A has gathered advantages and disadvantages. Although BPA is the crucial factor in manufacturing baby bottles, it was inevitable that there would be impact of Bisphenol A in baby bottles on children health. The negative effects associate with prevalent diseases in modern life such as early puberty and obesity. Hence, governments and scientists take into action as soon as possible to preclude child health expenditures, which are closely related to large economic burden. In the future, it is necessary to test potential substitutes in order to prevent the use of replacement Bisphenol A that may lead the same or worse health consequences.
REFERENCES
- Calafat A.M., Ye X., Wong L.Y., Reidy J.A., and Needham L.L., “Exposure of the U.S. Population to Bisphenol A and 4-tertiary-octylphenol: 2003-2004,” Environ Health Perspect, 2008 Jan;116(1):39-44
- Bisphenol A. (2005). [online] Available at: http://pubchem.ncbi.nlm.nih.gov/compound/Bisphenol_A [Accessed 2 Apr. 2015].
- Hc-sc.gc.ca, (2015). ARCHIVED – Survey of Bisphenol A in Canned Drink Products [Health Canada, 2009]. [online] Available at: http://www.hc-sc.gc.ca/fn-an/securit/packag-emball/bpa/bpa_survey-enquete-can-eng.php [Accessed 7 Apr. 2015].
- Karin B, M. (2015). BPA, chemical used to make plastics, found to leach from polycarbonate drinking bottles Into humans | News | Harvard T.H. Chan School of Public Health. [online] Hsph.harvard.edu. Available at: http://www.hsph.harvard.edu/news/press-releases/bpa-chemical-plastics-leach-polycarbonate-drinking-bottles-humans/ [Accessed 11 Apr. 2015].
- Corvese, G. (2013). BPA linked to obesity in young girls. [online] Thechart.blogs.cnn.com. Available at: http://thechart.blogs.cnn.com/2013/06/12/bpa-linked-to-obesity-in-young-girls/ [Accessed 11 Apr. 2015].
- Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of childhood and adult obesity in the United States, 2011-2012.Journal of the American Medical Association2014;311(8):806-814.
- Harrington, R. (2010). Sugar-derived epoxy lining could replace bisphenol A. [online] FoodProductionDaily.com. Available at: http://www.foodproductiondaily.com/Packaging/Sugar-derived-epoxy-lining-could-replace-bisphenol-A [Accessed 11 Apr. 2015].
- Empen, K. (2013). The Facts About Bisphenol A, BPA. [online] Webmd.com. Available at: http://www.webmd.com/children/environmental-exposure-head2toe/bpa [Accessed 11 Apr. 2015].
- Niehs.nih.gov, (2015). Bisphenol A (BPA). [online] Available at: https://www.niehs.nih.gov/health/topics/agents/sya-bpa/ [Accessed 11 Apr. 2015].
- Trasande, L. (2014). Further Limiting Bisphenol A In Food Uses Could Provide Health And Economic Benefits. Health Affairs, 33(2), pp.316-323.
- Fda.gov, (2010). Bisphenol A (BPA): Use in Food Contact Application. [online] Available at: http://www.fda.gov/NewsEvents/PublicHealthFocus/ucm064437.htm [Accessed 11 Apr. 2015].
- Vom Saal et al., “Chapel Hill Bisphenol A Expert Panel Consensus Statement: Integration of Mechanisms, Effects in Animals and Potential to Impact Human Health at Current Levels of Exposure,” Reproductive Toxicology, 2008 August-September, pp. 131-138.
1
Title: Impact of Bisphenol A in Bottles on Children’s health
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allDMCA / Removal Request
If you are the original writer of this essay and no longer wish to have your work published on UKEssays.com then please click the following link to email our support team:
Request essay removal


