Distillation of Crude Oil in Petroleum Refining
| ✅ Paper Type: Free Essay | ✅ Subject: Chemistry |
| ✅ Wordcount: 1789 words | ✅ Published: 23 Sep 2019 |
Wiktoria Swiatek
K1843100
COURSEWORK ASSIGNMENT
Chemistry SX3003
Section 1: Petroleum Refining
1a) Process of distillation of crude oil in Petroleum Refining
Petroleum is classified as a fossil fuel. Fossil fuels take millions of years to form; therefore petroleum is considered a non-renewable energy resource. Current estimations tell us that within the next few decades, this resource will no longer be available.
Petroleum refining is the process of converting crude oil into useful products for industrial usage and its necessities used on a day-to-day basis, such as Gasoline, Diesel, Fuel Oil etc. During the process of Petroleum Refining, the hundreds of hydrocarbons in crude oil are separated and converted into these useful products. (Organic Chemistry, John E. McMurry).
Petroleum Refining is shown through three different stages; separation, conversion and treating. Separation is where the distillation process occurs. Distillation is when condensation occurs through the use of thermal energy for liquids. The heated crude oil enters a tall fractionating column, which (as the diagram shows, it’s warmer at the bottom- reaching 400 °C and cooler at the top, starting at 20 °C). The crude oil is heated by a furnace, in which the hydrocarbons can be separated into different boiling points. The lighter fractions reach the top of the tower where they condense into a liquid state; here they’re at a low boiling point. The heavier fractions stay at the bottom of the tower and have a high boiling point. Each of the fractions contain hydrocarbon molecules and a similar amount of carbon atoms. (Petroleum.co.uk – accessed – December 2018)
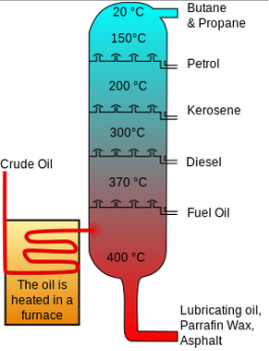 (Figure 1 Diagram of a Fractional Distillation Tower) (Energyeducation.ca – accessed – January 2019)
(Figure 1 Diagram of a Fractional Distillation Tower) (Energyeducation.ca – accessed – January 2019)
This diagram shows where the different fractions will condense. The temperature is higher at the bottom, the longer carbon chains will fall out at the bottom, the shorter carbon chains will go up the column until they hit the high temperature.
|
Petroleum fractions |
Boiling point Range |
Uses |
|
Gases |
0C-30C |
Gases can be bottles and used for industrial use, it is also found as a ‘natural gas’ |
|
Napthas |
30C-260C |
Petrol |
|
Kereosenes |
180C-260C |
Kereosene is used as a jet fuel and I it is found in home heaters. |
|
Gas oils |
260C–350C |
Gas oils can be used as disel fuels, and is used in the the process of ‘crackling’ |
|
Lubicants |
350C-574C |
Lubricants can be used in car oil and in the process called ‘crackling’ |
|
Fuel oil |
490C |
Fuel oil can be used in candles and in power stations. |
|
Asphalt |
580C |
Asphalt used in road construction in the form of tar, in addition it is used in the form of roof tar. |
1b) Describe the process of catalyst cracking in Petroleum Refining
The catalyst cracking happens during the second stage of petroleum refining. This process is needed because the heaviest fractions including gas oil and residual oil are lower in value than some of the other lighter fractions. This process is used to break apart the molecules in these heavier fractions. Through this, the end products will have a higher value. Jet fuel and gasoline are produced from the utilization of cracking that are initially heavy gas oils. (www.e-education.pdsu 2019)
Catalytic cracking is used in petroleum refining to break down long chains of hydrocarbons into simpler molecules (smaller hydrocarbons) by breaking the ‘carbon-carbon’ bonds. Large hydrocarbons are vaporised and placed over a hot catalyst. The chemical bonds within the hydrocarbons are broken down by the heat, this makes them smaller and so they’re much more efficient for during the usage (also known as thermal cracking). Catalyst cracking is a necessity in the process of Petroleum Refining as using large hydrocarbons within oils is not an efficient way to produce fuels. So by breaking it down into smaller alkanes and alkenes, it’s more useful during the production of fuels and alkanes specifically in the process of making polymers. (BBC Bitesize – accessed December 2018)
Example of catalytic equation (BBC Bitesize – accessed December 2018) :
hexane → butane + ethene
C6H14 → C4H10 + C2H4
Butane – gas
Ethene – gas
Hexane – liquid
From my research , I know that Hexane can be cracked under a high pressure and high temperature of butane and ethane.
Butane uses: Liquid petroleum gas, Petrol, Petrochemicals, Fuel, deodorant sprays etc.
Ethene: Polyethene, PVC, Chloroethene, Ethanol (John E. McMurry and BBC Bitesize, 2019)
Section 2 – Buffers :
A Buffer solution is an environmental solution, which resists the specific changes in the pH when either an Alkaline or Acid is added to it (Chemguide, accessed – 2019)
Examples of Buffer solution used on a day-to-day basis: Inside the human body, the main buffer that is in use, is in the blood stream. Carbonic Acid and Hydrogen Carbonate work together to maintain a dynamic equilibrium in the body to disallow a very acidic or alkaline solution. They maintain the pH and temperature too so cells in the mammalian body do not denature the body system failures. Another example in the body, is that the buffer solutions are used in the skeletal muscles during exercise – the Haemoglobin binds to specific proteins during contraction and dilation. With things that are used by humans; a buffer is used to balance out the alkalinity in the soap of a shampoo. During the fermentation of alcohol, a buffer is used to maintain the pH and thus produce a maximum yield of alcohol. And in the manufacturing of glue, some ingridients inside it can change the pH within solution change, the buffer here is used to withstand the changes during the manufacturing of glue. (chemsitewebsite,weebly.com – accessed in 2019)
The Common types of buffers are Acetic acid + sodium acetate and Citric acid + sodium citrate.
Common buffer solutions, for example : Acetic acid + sodium or acetate Citric acid + sodium citrate. (John E. McMurry, 2017 and Chemguide, 2019)
A weak acid: A weak acid is a chemical solution which cannot fully dissociate with water. Meaning the hydrogen ions cannot fully bind to the water molecules. This can be presented by H3COOH(aq) ⇌ CH3COO– (aq) + H+ (g) – (John E. McMurry, 2017 and ThoughtCo, 2019)
The weak acid aspirin is partially dissociated in an aqueous solution with sodium salt.
HAsp(aq) ⇌ asp-(aq)- + H+(aq)
The buffer solutions works when an acid (such as weak acid aspirin) is added to the new hydrogen ions and have to be fully removed from the solution in order to resist a change in pH. This happens when the aspirin is added to the solution, so that the hydrogen ions are removed from the solution. (Chemguide.co.uk, 2019)
Asp-(aq) + H+ ⇌ Hasp(aq)
– When the base is added to sodium salt-
Hasp(aq)+OH-(aq) ⇌asp-(aq)+H2O .
Here we can see that the newly added hydroxide ions OH are removed by the ‘Hasp’. This forms asp and H20, this means that the pH would remain constant. (Chemguide.co.uk, 2019)
Section 3 : Bond Energy and Hess’s Law
1a)
2C(s) + 3H2(g) → C2H6(g)
 2C(s) + 3H2(g) C2H6(g)
2C(s) + 3H2(g) C2H6(g)



3(-286)
2(-394) 1560
 2CO2(g) + 3H2O(l) C2H6(g)+ 3½O2(g)
2CO2(g) + 3H2O(l) C2H6(g)+ 3½O2(g)
(2x-394) + (3x-286) = -1646
1560 – 1646 = – 86kjmol-1
1b)
 2C(s) + 3H2(g) C2H6(g)
2C(s) + 3H2(g) C2H6(g)
 2(717)+3(436) -346+6(-412)
2(717)+3(436) -346+6(-412)
2742 – 2818 = -76kJmol-1
1c) There is a discrepancy of 10kJmol-1. The value from section (a) is more likely to be accurate as this data was collected from an experiment, however the average bond enthalpies used in section (b) are a standard.
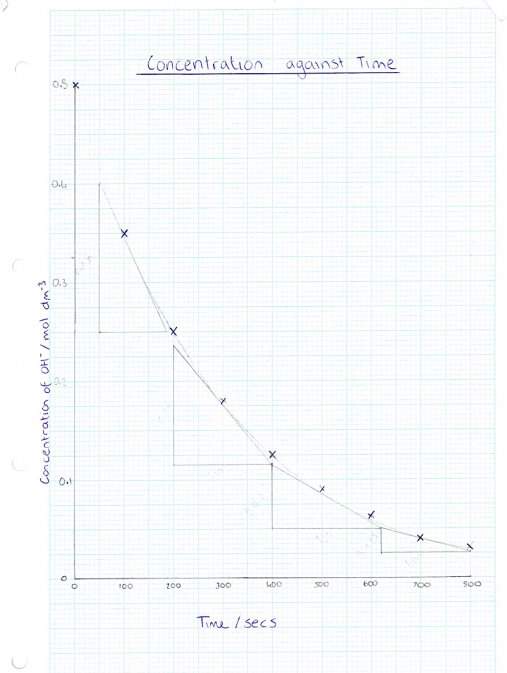
Section 4: Rates of Reaction
4b)
|
Rate |
Concentration |
|
0.35 |
0.001 |
|
0.175 |
0.0006 |
|
0.075 |
0.000295 |
|
0.03 |
0.000138 |
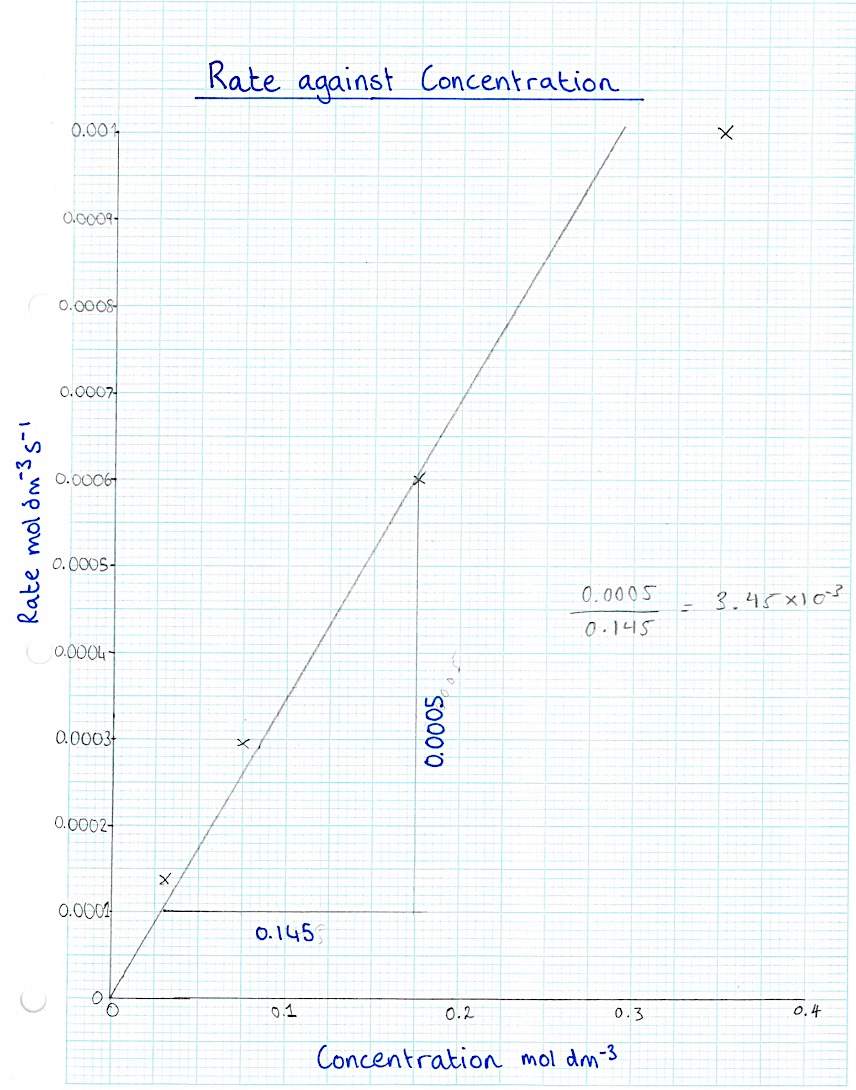
4d) The order of reaction is in the form : y=mx+c (which is the equation for a straight line). As the graph goes through the origin, c = 0 and m is the gradient, in this case k. (Kingston university resources)
4e)
4f)
REFERENCES:
- Organic Chemistry 7th Edition (2007) Distillation: Author, John E. McMurry, Organic Chemistry, Cengage Learning.
- Petroleum.co.uk. (2019). Petroleum – Refining. [online] Available at: http://www.petroleum.co.uk/refining [Accessed 17 Dec 2018].
- Diagram of distillation tower – https://energyeducation.ca/encyclopedia/Fractional_distillation (accessed January 2019)
- BBC Bitesize. (2019). GCSE Chemistry (Single Science) – Crude oil, hydrocarbons and alkanes – AQA – Revision 5. [online] Available at: https://www.bbc.com/bitesize/guides/zshvw6f/revision/5 [Accessed 20th Dec. 2018].
- Organic Chemistry 7th Edition (2007) Distillation: Author, John E. McMurry, Organic Chemistry.
- What is distillation? https://www.britannica.com/science/distillation (accessed on January 2nd 2019)
- What is the process of a catalyst cracking? https://www.e-education.psu.edu/eme801/node/470 (accessed on the 2nd January – 2019)
- Chemguide.co.uk. (2019). buffer solutions. [online] Available at: https://www.chemguide.co.uk/physical/acidbaseeqia/buffers.html [Accessed 16 Jan. 2019].
- Every day buffers – http://chemsitewebsite.weebly.com/-buffers.html (Accessed on the 17 Jan 2019)
- ThoughtCo (2019) what is a weak acid? Available at: https://www.thoughtco.com/definition-of-weak-acid-604687 (Accessed January – 2019)
- Hess’s Law cycle – https://www.chemguide.co.uk/physical/energetics/sums.html (Accessed january 2019 )
- Chemistry Libretexts (2019) Thermochemistry. Available at: https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Map%3A_Physical_Chemistry_for_the_Biosciences_(Chang)/03%3A_The_First_Law_of_Thermodynamics/3.6%3A_Thermochemistry (Accessed January 4th 2019)
- Order of Reaction – Kingston Resources- www.canvas.uk (accessed on January 23rd 2019)
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allDMCA / Removal Request
If you are the original writer of this essay and no longer wish to have your work published on UKEssays.com then please click the following link to email our support team:
Request essay removal


