Catalase Kinetics with the Decomposition of Hydrogen Peroxide with Temperature Changes
| ✅ Paper Type: Free Essay | ✅ Subject: Chemistry |
| ✅ Wordcount: 839 words | ✅ Published: 23 Sep 2019 |
Examining Catalase Kinetics with the Decomposition of Hydrogen Peroxide with Temperature Changes
Table of Contents
1.1 Introduction
1.1.1 Importance of Experiment
1.1.2 Purpose
1.1.3 Hypothesis
1.1.4 Prediction
1.2 Results
1.2.1 Statement
1.2.2 Figure
1.1 Introduction
1.1.1 The Importance of Studying Catalase
The body expends valuable energy to compensate for external pressures that cause stress on the physical equilibrium in the body. Naturally, under stress, the body will increase the concentration of reactive oxygen species (e.g., hydrogen peroxide) from oxidative metabolism. Disruption of the body’s tendency to maintain ROS homeostasis may lead to oxidative stress damage and chronic adverse effects (Halliwell B., 1998). However, cells reduce this risk by weaponizing antioxidizing enzymes like catalase that catalyzes the decomposition of hydrogen peroxide into water and oxygen. Like other cellular enzymes, an enzyme’s efficiency depends on variables like pH, temperature and inhibition. Enzyme inhibition or inactivation of catalase can lead to damage to macromolecules, including nucleic acids, and cell death by apoptosis or necrosis (Halliwell B., 1998).
1.1.2 Purpose
In this experiment, the goal was to determine whether catalase activity regarding the decomposition of hydrogen peroxide was affected by an increase in temperature (22oC vs. 37oC).
1.1.3 Hypothesis
The induced activity of catalase will increase if temperature increases.
1.1.4 Prediction
If catalase activity increases with temperature, then indirectly measuring the pressure rate of released molecular oxygen should prove whether this relationship is true.
1.4 Result
1.4.1 Statement and 1.2.2 Figure
The Experimental Group (n=12) had a significantly higher (p = 9.411e-6) production rate of molecular oxygen than the Control Group (n=12; Figure 1).
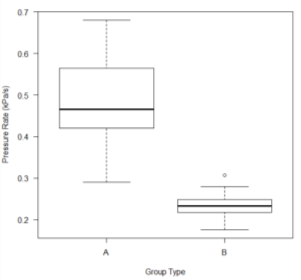
Figure 1. The pressure rate (kPa/s) of the amount of molecular oxygen released I observed from each group. Group A is the Experimental Group exposed at thirty-seven degrees Celsius. Group B is the Control Group exposed at twenty-two degrees Celsius.
1.3 Annotated Bibliography
Cantemir, A. R., A. Raducan, M. Puiu, and D. Oancea. 2013. Kinetics of thermal inactivation of catalase in the presence of additives. Process Biochemistry 48:471–477.
- In 2013, researchers Cantemir, A. et al. questioned how temperature and the presence or absence of additives and substrate affect the kinetics and temperature inactivation of mammalian catalase (using incubation and iso conversional methods). Substrate concentrations were calculated using Lambert-Beer Law. The researchers concluded that the inactivation of catalase was significantly larger when hydrogen peroxide was present. When the substrate was absent, the initial rate of reaction for catalase with additives decreased exponentially with temperature increase. The team observed that the maximum for catalase activation with water and substrate was forty degrees Celsius. This study holds valuable information about the kinetics of catalase. This study will provide insight into how we can predict, understand and interpret the effects of temperature (independent variable) on the rate of catalase activity.
Fidaleo, M., and R. Lavecchia. 2003. Kinetic study of hydrogen peroxide decomposition by catalase in a flow-mix microcalorimetric system. Thermochimica Acta 402:19–26.
- In Thermochimica Acta, researchers examined the decomposition kinetics of hydrogen peroxide by catalase. The reactions were observed at a pH of 7.4 and within a temperature range of ten to thirty degrees Celsius in a four-channel isothermal microcalorimeter. Using the data, researchers calculated the thermodynamic quantities of the reactions. First, the team calculated the enthalpy of reaction, found at -87.55 ± 0.72 kJ/mol, then the Gibbs Energy at twenty-five degrees, calculated at -119 kJ/mol. The rate constant, k, was determined by devising a function, k(T), with experimental values of activation energy and kinetic parameters. Once plotted, it was apparent that an increase in temperature increased the rate constant. As stated in the study, a high rate constant indicates a spontaneous reaction, due to the low activation energy. Thus, within the temperature range in this experiment, the rate of the decomposition reaction catalyzed by catalase increased with temperature. This study proved that this reaction is spontaneous and exergonic with the help of the enzyme. The laws of thermodynamics and entropy favor this reaction. The characteristics of this reaction are relevant as this reaction is nearly identical to the reaction I conducted when observing the kinetics of catalase based on the rate of gas production. As well, this study provides the foundational knowledge for understanding the nature of this reaction.
Halliwell, B. 1998. Biomarkers of oxidative damage associated with human disease. Pathophysiology 5:267.
Nannenga, B. L., D. Shi, J. Hattne, F. E. Reyes, and T. Gonen. 2014. Structure of catalase determined by MicroED. eLife 3.
- Using electron diffraction (MicroED), researchers at Howard Hughes Medical Institute in 2014 determined the three-dimensional structure of catalase. They were the first to do so with this method. The data was processed and refined in X-Ray crystallography programs. The study’s results reinforced the current understanding of catalase structure. Catalase consists of monomers with heme groups, bound to an NADP molecule, with variable thickness of protein layers. The unit cell is asymmetrical each with four molecules. The study additionally plotted the ligand and paired conformational changes in ligand-binding regions. The structure of catalase predetermines its function. Hence, this study provides an idea of how this enzyme’s function is reduced or increased because of how temperature can alter its shape (binding regions).
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allDMCA / Removal Request
If you are the original writer of this essay and no longer wish to have your work published on UKEssays.com then please click the following link to email our support team:
Request essay removal


