Antiglycation Studies of Pd(II)-Hydrazide Complexes
| ✅ Paper Type: Free Essay | ✅ Subject: Chemistry |
| ✅ Wordcount: 2168 words | ✅ Published: 18 Jan 2018 |
- Introduction
Glycation is a non-enzymatic spontaneous reaction between sugars and coexisting protein followed by a complex cascade of reactions including dehydration, redox reaction and other rearrangements [1, 2] forming advanced glycation end products (AGEs) [3]. Glycation reactions depend on the generation of reactive oxygen species by trace amounts of redox active metal ions [4] and on the degree and duration of hyperglycemia in vivo [5]. Glycation damages the collagen and elastin throughout the body. It is generally accepted that accumulation of tissue AGEs together with enhanced oxidative stress has an important role in the progression of aging and diabetic complications including retinopathy, neuropathy, embryopathy, delayed healing of wounds and others [6-10]. The increase in diabetic complications is the major cause of increased morbidity and mortality rate that has enhanced considerably in the two decades [11]. It has been estimated that the number of cases of DM will reach to 366 million by 2030 [12, 13] showing in fact a great challenge to healthcare systems [14]. The failure of existing antidiabetic drugs are forcing researchers to find out new inhibitors of proteins responsible for glycation in order to have a long term and sustainable solution for management of diabetes and age-related diseases.
Protein Glycation
The protein glycation, also called Millard reaction, involves non-enzymatic coupling of proteins with reducing sugars eventually producing advanced end products. The glycation is a spontaneous reaction, which stimulates the degradation of proteins with modification of their structures and biological activity [1-3]. Various reducing sugars including glucose, lactose, fructose, xylose, deoxyribose and galactose may take part in protein glycation [15].
Chemistry and Mechanism of Protein Glycation
The protein glycation process initiates with the reaction of carbonyl (keto or aldehydic) group of reducing sugar with free amino group of protein forming a labile Schiff base 16. This is called early stage of glycation. The Schiff bases are then transformed through Amadori rearrangement into comparatively stable compounds known as Amadori products. At acidic pH or under oxidative conditions, the Amadori products or Schiff bases undergo degradation generating extremely reactive 1, 2-dicarbonyl compounds, such as methylglyoxal (Figure 94) 17-21. The formation of protein dicarbonyls through a protein enediol may generate superoxide radicals in the presence of transition metal ions and molecular oxygen 22. The superoxide radicals can be converted into most reactive hydroxyl radical via Fenton reaction 7. The reactive carbonyl compounds subsequently react with amino groups of neighboring proteins producing protein dicarbonyl compounds, which further contribute in the formation of various types of protein crosslinks and adducts called ‘Advanced Glycation End Products’ (AGEs). The autoxidation of Amadori products to AGEs is described as glycoxidation process.
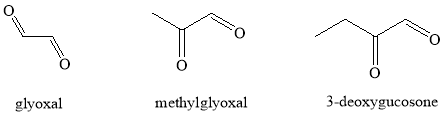
Figure 94. Structures of some reactive dicarbonyl glycation intermediates
Advanced Glycation End Products
The advanced glycation end products (AGEs) comprise a complex heterogeneous group of compounds produced primarily through the reaction of reactive carbonyls and proteins. AGEs demonstrate to have diverse molecular biological functions and structures 18, 23]. The amino, sulphydryl and guanidinum functional groups occurring in the intracellular and extracellular proteins are the main targets of reactive carbonyl compounds. Various AGEs have been recognized in different tissues that can be categorized into three major groups: fluorescent cross-linked AGEs (e.g. pentosidine and crossline), non-fluorescent cross-linked AGEs (e.g. alkyl formyl glycosyl pyrrole and arginine-lysine imidazole cross-links) and non-cross linked AGEs (e.g. pyrraline and N-(carboxylmethyl) lysine) 18. The structures of some AGEs are presented in Figure 95.

Figure 95. Structures of selected advanced glycation end products
Factors Affecting the Formation of AGEs
In physiological environment, the generation of AGEs is a relatively slow process. Accordingly, the AGEs accumulation is dominant in long-lived structural proteins, for instance, tissue collagens and lens crystallins. The oxidative conditions are known to accelerate the formation of AGEs, which slows down under anaerobic environment [24]. The transition metal ions may induce the auto-oxidation of sugars to produce keto aldehydes and hydrogen peroxide that speed up the formation of AGEs [25]. The amount of AGEs formed is increased as a function of time and concentration of glucose and hence the AGEs formation is enhanced with aging and under diabetic conditions [26].
Site Specificity of Glycation of Proteins
Glycation of protein is considered as a specific reaction; however, it is less specific compared to enzymatic glycosylation. Glycation often takes place at specialized site in the protein, such as the substrate binding site (e.g. Arg-39) of RNase, the allosteric site (e.g. βV1) of hemoglobin, and the drug binding sites (e.g. Arg-410) on albumin [27]. The specificity of glycation may be determined by endogenous ligands and the structure of protein especially an amino acid sequence within the protein. The specificity of protein glycation is usually affected by both basic and acidic neighboring groups [28], either via catalysis of Amadori rearrangement (the rate-limiting step of protein glycation), or via effecting pKa of amino group that contributes in enhancing its nucleophilicity and formation kinetics of Schiff bases. This shows that there is a variation in the respective rate and extent of glycation shown by amino groups in the protein. The anionic ligands also catalyze the potential glycation of proteins at specific sites [27].
Exogenous Sources of AGEs
The formation of AGEs via Maillard reaction was originally described for physical and chemical changes occurring during heating of food [29]. Beside the natural formation of AGEs inside the body, there are some exogenous sources of increased AGEs including diet enriched with AGEs and smoking. The extent of absorption of AGEs ingested with food is very small [30]. However, there is a strong relationship of AGEs circulating in the human body with the AGEs ingested [31].
It has been investigated that tobacco smoke increases the formation of AGEs on plasma proteins due to containing some products, which produce protein crosslinks and AGE-like fluorescence and mutagenicity [32]. For example, the diarbonyl compounds, glyoxal and metbylglyoxal, are most likely to be present in cigarette smoke that act as mediators of AGE formation and formed by thermal decomposition of existing saccharides. Accordingly, the serum of diabetic smokers reveals enhanced levels of AGEs relative to diabetic non-smokers [33]. Similarly, the smokers are more susceptible to incidence of cataract, cardiovascular and lungs diseases as compared to non-smokers due to smoke-mediated AGEs formation [34].
Toxicity or Pathological Conditions Associated to Glycation and AGEs Formation
The AGEs are more prone to proteolysis and degradation as compared to the original proteins. The accumulation of AGEs has toxic biological effects, causing disruption of many cellular processes leading to various pathologies. The AGEs as well as intermediate glycation products such as reactive carbonyls induce the production of free radicals in vitro and in vivo [35, 36] and hence increased oxidative stress [37]. The glycation-mediated free radicals are the major cause of protein fragmentation as well as oxidation of lipids (lipid peroxidation) and nucleic acids [7].
The reactive dicarbonyls have ability to bind with naturally active proteins of diverse physiology via intra- and inter-molecular cross linking resulting in deactivation of enzymes, transcription factors, membrane transporters and signaling components with eventual protein degradation and cytotoxicity [38-40]. The AGEs also bind to cell membrane receptors inducing signal cascades leading to inappropriate gene expressions and cellular activities [18].
The elevated level of AGEs in tissues has a strong correlation with severity of diabetic complications [41, 42]. This is because of modification of enzymatic activity in multiple ways including binding of ligand, change in protein half life, increased membrane permeability, decreased binding ability of insulin to its receptors, increased atherogenicity of LPL and variation in the immunogenicity [43-45]. The main diabetic complications include impaired wound healing and the serious damage and failure of various vital organs such as kidneys (nephropathy), nerves (neuropathy), eyes (cataract, retinopathy), blood vessels (atherosclerosis) and heart (cardiomyopathy) [11, 34, 46, 47]. The AGEs formation is also associated to aging, Alzheimer’s disease and other chronic disorders [17, 10, 48-50].
Natural Biological Defense Mechanism against Glycation and AGEs
The human body presents a certain mechanism to inhibit the glycation of protein and resulting AGEs formation. For example, α-keto-glutaraldehyde dehydrogenase, a liver enzyme, has a capability to inactive 3-deoxyglucosone (3-DG), Arnadori-derived reactive intermediate and hence prevents the generation of AGEs [41]. The other enzymes such as aldose reductase and glyoxylase system (I and II) can catalyze the deglycation of reactive intermediate methylglyoxal into D-lactase [51]. Amadoriases are the group of enzymes found in Aspergillus, which catalyze the deglycation of Amadori products [52]. Some NADPH-dependant exogenous enzymes such as aldose reductase and oxoaldehyde reductase that metabolize α-dicarbonyls, have the ability to reduce 3-DG and thus regulate the formation of AGEs [53]. Similarly, different plasma amines may reduce AGEs formation through reaction with carbonyl groups of sugar and Amadori compounds [54]. Antioxidants such as vitamin E and vitamin C, provide protection against glycation-mediated free radicals, whereas, ceruloplasmin and other transport proteins bind with transition metal ions such as Cu2+, preventing them to take part in glycoxidation reactions or autoxidative glycation [55].
Inhibition of Protein Glycation and AGEs
Several attempts have been made earlier to explore pharmacologically active antiglycating agents to prevent or slow down the production of AGEs [56]. The major side effects associated with antiglycation therapy limit the use and necessitate the discovery of new inhibitors of glycation with reduced toxicity and long half life to be implicated for large time span. Currently, two therapeutic strategies are highly successful having great effectiveness against diabetic complications and normal aging; one is the inhibition of formation of AGEs and other is the breaking of already established AGEs cross-linkages [57].
Promising Inhibitors of Glycation with Their Mechanism of Inhibition
The antiglycating agents such as aminoguanidine, rutin, antioxidants, aspirin and other AGEs breakers have been examined extensively and received great interest. The structures of some potential antiglycating agents are depicted in Figure 96.
Aminoguanidine
Aminoguanidine is a derivative of hydrazine that inhibits the generation of AGEs and glucose-derived collagen cross-links during in vitro studies [58]. Aminoguanidine does not act on already formed AGEs but it reacts with reactive Amadori products such as 3-deoxyglucosone preventing additional rearrangements and intermediates crosslinking [59]. In addition, aminoguanidine is a free radical scavenger that contributes in reducing oxidative stress [60]. The treatment of diabetic animal models with aminoguanidine reduces AGEs accumulation, kidney lesions, albuminuria and long-term diabetic complications including retinopathy, nephropathy and neuropathy [61]. Aminoguanidine also exerts positive effect on the speed of nerve conduction [22]. Aminoguanidine therapy is limited by serious toxic effects attributable to high reactivity, subliminal concentrations and rapid renal clearance. The human trials with aminoguanidine experience vasculitis (inflammation of lymph or blood vessel), liver function abnormalities [62] and less frequent flu-like symptoms, nausea and headache [63].

Figure 96. Structures of some potential antiglycating agents
Aspirin
Acetylsalicylic acid commonly known as aspirin is an analgesic has well known analgesic drug that also shows the preventive action against formation of cataract under diabetic conditions. Aspirin may limit the sugar-mediated formation of Amadori products by acetylation of free amino residues of proteins. Aspirin also stops the crosslinking of tendon in rat tail in vitro through inhibition of glycoxidation. Furthermore, aspirin is a free radical scavenger [64]. However, the use of aspirin is unlikely in controlling late diabetic complications because of some serious gastrointestinal side effects [41].
Rutin
Rutin is a common flavonoid of vegetables and fruits that modulates the AGEs generation in vitro. The flavonoids including rutin that contain vicinal dihydroxyl groups have established their significant role as antiglycating agents. The mechanism of inhibition by rutin is suggested to involve the trapping of amino groups in proteins at early stage of glycation, especially in ketoamine formation, by rutin metabolites like keto-quinone intermediates. Rutin has shown significant inhibitory effect against hemoglobin glycation and it is more efficient compared to aminoguanidine [65].
Antioxidnats
Since non-enzymatic glycation of protein is significantly accelerated by excessive generation of free radicals, the antioxidants and other free radical scavengers are expected to inhibit the process of glycation [24]. For example, vitamin E has been reported to appreciably reduce the glycation of hemoglobin [66]. The compounds that exhibit both antioxidant and antiglycation properties e.g. aminosalicylic acid, can protect endothelial cells with better efficacy than aminoguanidine against adverse effects of glycation and high glucose levels in vitro [46]. Similarly, carnosine that is a naural antioxidant and antiglycating agent, inhibits sugar-induced cross-linking of proteins by reaction with methylglyoxal and also sequesters metal ions (e.g. copper and zinc). Carnosine has shown its role in the treatment of cataracts and other diseases [67]. The glycation-derived free radicals generation may be reduced by chelation of transition metal ions, which are responsible for monosaccharide autoxidation. For instance, the metal chelator, diethylenetriamine penta acetic acid has shown the inhibition of glucose autoxidation [68, 69].
AGE-Breakers
AGE-breakers are the compounds, which remove AGEs cross-links through breakdown of α-dicarbonyl bonds in glucose-derived cross-links of proteins [70]. However, the exact mechanism of inhibition for cross-link breaking is unclear so far. N-phenacylthiazolium bromide (PTB) and its chloride form, alagebrium chloride (ALT-711) are the examples of AGE-breakers. It has been demonstrated that the increased arterial stiffness related to diabetes is successfully reversed through a short treatment with AGE-breaker, ALT-711. The cardiovascular stiffness related to normal aging process can also be reduced by ALT-711. For example, the treatment of normal aged dogs with AGE cross-link breaker has shown noticeable decrease in stiffness of left ventricle chamber [71].
The clinical trial studies on diabetic humans, dogs and other animals indicate the potentially promising use of antiglycation therapy in near future to prevent diabetic complications and other diseases related to protein glycation [56].
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allDMCA / Removal Request
If you are the original writer of this essay and no longer wish to have your work published on UKEssays.com then please click the following link to email our support team:
Request essay removal


