Haemolytic Disease of the Fetus and Newborn (HDFN)
| ✅ Paper Type: Free Essay | ✅ Subject: Biology |
| ✅ Wordcount: 2912 words | ✅ Published: 12 Jun 2018 |
List and briefly describe three clinical signs for Haemolytic Disease of the Fetus and Newborn (HDFN)
Haemolytic disease of the fetus and newborn (HDFN) is a rare disease that occurs when maternal alloantibodies cross the placenta during pregnancy and cause the destruction of fetal red blood cells (RBCs) (Delaney and Matthews, 2015; Haas et al., 2015). HDFN can result in fetal anaemia with progression to severe morbidities, such as ascites, hydrops fetalis, heart failure, kernicterus, and death (Delaney and Matthews, 2015). The clinical presentation of HDFN is variable, in which there are several manifestations that may occur (Murray and Roberts, 2007). Three of the most prevalent clinical signs that allows for neonatal paediatricians to suspect HDFN includes splenomegaly, oedema, and jaundice.
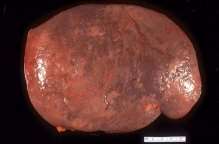

HDFN is characterised by the accelerated destruction of RBCs, which results in differing rates of haemolysis and fetal anaemia (Urbaniak and Greiss, 2000). The continuous and rapid process of haemolysis causes extramedullary haematopoiesis, a result of erythropoiesis failing in the bone marrow, in the fetal liver and spleen (Dean, 2005). Organs, such as the liver and spleen, that are involved in the synthesis of RBCs increase the production to combat the rate of destruction and counteract the overall loss (Dean, 2005). The increased workload of the spleen results in its enlargement, termed splenomegaly (Bowman, 1997; Dunn, 1963).
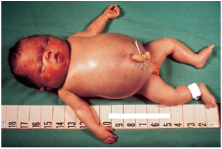

Oedema is an important clinical sign of HDFN that is also associated with the degree of haemolysis and anaemia (Delaney and Matthews, 2015). Oedema can occur due to low levels of serum albumin through a decrease in osmotic pressure (Dean, 2005). Moreover, as the body compensates for fetal anaemia, the fetus can develop a hyperdynamic circulation (Haas et al., 2015). This can result in hydrops fetalis, a severe and life-threatening condition in which there is widespread oedema in the fetal and skin and serous cavities (Haas et al., 2015).
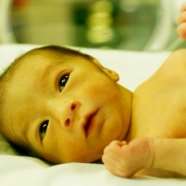

A further clinical sign of HDFN is jaundice. Jaunice may occur as a result of haemolysis, in which there is an increase in the level of bilirubin within the body (Urbaniak and Greiss, 2000). Throughout pregnancy, bilirubin is removed via the maternal circulation by the placenta (Dean, 2005). Therefore, a high level of haemolysis may be present with a low level of bilirubin (Murray and Roberts, 2007). However, after birth the haemolytic process continues. At this stage of development, the liver of the neonate is immature and unable to conjugate the excess bilirubin (Urbaniak and Greiss, 2000). The unconjugated bilirubin begins to build and accumulate in the blood of the neonate, causing the skin and whites of the eyes to turn yellow (Dean, 2005). Within 24 to 48 hours after delivery, the level of bilirubin may increase substantially (Urbaniak and Greiss, 2000). Left untreated, this can lead to the development of kernicterus, a condition in which bilirubin deposits form in the basal ganglia and brain stem nuclei (Haas et al., 2015).
Describe the progression of HDFN, from sensitisation to fetal red cell destruction, in a D negative mother carrying a D positive fetus that has not received RhIg.
Throughout pregnancy, antibodies from the maternal circulation play a vital role in providing protection for neonates; crossing the placenta from the maternal to the fetal circulation (Dean, 2005). This is essential for the fetus, as by delivery newborns have a relatively immature immune system (Murray and Roberts, 2007). Although the presence of maternal antibodies provides protection, the active transporting of antibodies across the placenta can result in HDFN (Dean, 2005).
HDFN is triggered by a sensitisation event. This most often occurs during the first pregnancy; however, it can also arise from a blood transfusion or organ transplant (Delaney and Matthews, 2015). During the course of pregnancy, the maternal and fetal circulations gradually mix with each trimester (Delaney and Matthews, 2015). This results in maternal alloimmunisation, as the maternal circulation has been exposed to foreign RBCs (Haas et al., 2015). Despite the relatively small amount of fetal blood that passes into the maternal circulation, only a small amount is needed for sensitisation to occur (Dean, 2005).
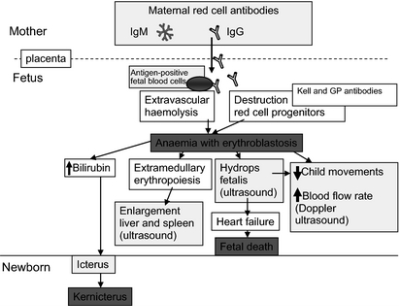

This is typical for an RhD negative mother carrying an RhD positive fetus. Sensitisation frequently transpires during the birth of the firstborn RhD positive child, where fetal maternal haemorrhage (FMH) is common (Delaney and Matthews, 2015). However, the risk of sensitisation increases in complicated and extensive labours (Dean, 2005). Sensitisation can also occur through earlier events in pregnancy, such as a prenatal bleed, trauma, termination of pregnancy, chronic villus sampling, and miscarriage (Sebring and Polesky, 1990). The greatest risk of developing FMH is during the process of labour (Murray and Roberts, 2007). Subsequently, alloantibodies are most likely to form after delivery (Delaney and Matthews, 2015). After sensitisation of an RhD negative mother carrying an RhD positive fetus, the mother’s serum will contain anti-D (Dean, 2005). Importantly, the maternal anti-D that is formed is of the IgM class and unable to effectively cross the placenta (Delaney and Matthews, 2015). As a result, HDFN is rare in first-born children and unlikely to have any clinical consequence or significance (Dean, 2005). However, once the maternal circulation has been exposed to the fetal circulation the maternal immune system has the potential to respond to foreign red cell antigens (Delaney and Matthews, 2015).
Through extensive research and cohort studies, it has been established that HDFN is most likely to effect subsequent pregnancies (Dean, 2005). Maternal alloantibodies of the IgG1 and IgG class cause significant haemolysis, thus the most clinically significant forms of HDFN (Roberts, 2008). In the event that an RhD negative mother becomes pregnant for a second time, interaction with the RhD antigen stimulates the production of IgG type anti-D, which can be transported across the placenta into the fetal circulation (Delaney and Matthews, 2015). Once anti-D has entered the fetal circulation, it binds to the RhD antigens found on fetal RBCs and labels them to be destroyed (Delaney and Matthews, 2015). From here, the pathophysiology of the disease ensues, as illustrated in figure 4.
Explain the action of RhIg in a D negative mother that has a D positive fetal bleed.
Rh immunoglobulin (RhIg) is routinely used in clinical practice to prevent HDFN. RhIg is prepared from human plasma that has been immunised to the D antigen and functions by targeting RBCs that are positive for the D antigen (Brinc and Lazarus, 2009). The use of prophylactic anti-D remains the gold standard approach of antibody-mediated immunosuppression, having been used for several decades (Giancarlo et al., 2010). However, the mechanism of action of RhIg is not fully understood and there are three key hypotheses that have been proposed to explain its method of action.
Antigen Clearance Hypothesis
The first hypothesis is the antigen clearance hypothesis and is considered the main mechanism of action. Here, IgG is understood to prevent an antibody response by increasing the rate of phagocytosis and the removal of RBCs from circulation via the mononuclear phagocytic system, prior to recognition by the immune system (Brinc and Lazarus, 2009). IgG opsonised RBCs are believed to engage in the activation of IgG receptors (FcγRs) on effector cells, stimulating phagocytosis. IgG is also suspected to increase the clearance of RBCs through the stimulation of complement activation on the RBC surface. Anti-D does not activate complement and therefore it is believed that FcγR-mediated phagocytosis is the mechanism by which anti-D is cleared (Brinc and Lazarus, 2009).
FcγRIIB mediated B-cell inhibition hypothesis
This mechanism is the most recently proposed and came about through the discovery of increased levels of transforming growth factor-β and prostaglandin E2 in a number of pregnant women who were given RhIG. This mechanism proposes that RBCs and IgG form a complex in which a negative signal is delivered to inactivate antigen-specific B cells. However, mice models deficient in FcγRIIBhave shown that the involvement of FcγRIIB is not needed to induce antibody-mediated immune suppression. Furthermore, FcR-like molecules have been discovered in both mice and humans. It is believed that the FCRLs mediate the B-cell inhibition, however, this has yet to be demonstrated (Brinc and Lazarus, 2009).
Steric Hindrance Hypothesis
The Steric hindrance hypothesis proposes IgG binds the antigen, preventing the B-cell receptor from recognising the corresponding epitopes. Most anti-D epitopes are not blocked by RhIg. This allows free D epitopes to be detected after administration of RhIg. Monoclonal anti-D has been shown to prevent antibody responses by binding 10-15% of epitopes. This pathway has not been studied in detail and therefore knowledge of the immunobiology is limited. However, it is believed that IgG binding of D epitopes allows a formation between RBCs and B cells, in which this prevents B-cell activation (Brinc and Lazarus, 2009).
The established methodology in the UK for the Quantification of a D positive fetal bleed is via flow cytometry with FITC-anti-D (FITC-BRAD3). Name and describe 3 alternative methods used worldwide to detect fetal bleeds
In addition to the use of flow cytometry, several alternative screening methods are available to determine and quantify FMH (Kim and Makar, 2011). Three screening methods that are used worldwide include the rosette screen, Kleihauer-Betke acid elution test, and flow cytometry using anti-fetal haemoglobin antibodies.
The rosette test is a screening method that is used to qualitatively detect fetal bleeds equal to or greater than 10 mL and 0.2% of fetal cells present in the maternal circulation (Kim and Makar, 2011). The rosette test works by indirectly identifying the presence of D positive fetal RBCs in D negative mothers (Solomonia et al., 2012). To perform this test, a maternal blood sample is collected, incubated with exogenous anti-D, and washed. D positive RBCs are added and are key as they act as an indicator. The sample is examined using a light microscope. In the presence of fetal D positive cells, the indicator RBCs form aggregates or ‘rosettes’ around the coated fetal RBCs (Solomonia et al., 2012).
A positive result is indicative of an FMH greater than 10 mL and requires quantification by Kleihauer-Betke acid elution test or flow cytometry to determine the dose of RhIg to administer (Kim and Makar, 2011). This test can encounter and produce false-positive results. This largely occurs if the mother of fetus is weak D. Furthermore, in the presence of a direct antiglobulin test (DAT), the rosette test may produce a false-negative result. This can be attributed to crosslinking and agglutination of the mother’s antibody coated cells (Kim and Makar, 2011).
The Kleihauer-Betke acid elution test is a screening method that differentiates between fetal haemoglobin (HbF) RBCs and adult Hb (Bromilow and Duguid, 1997). The underlying notion of this test is fetal RBCs largely contain HbF and are resistant to acid elution, whereas in contrast, adult Hb is acid-sensitive (Kim and Makar, 2011). To perform this test, a maternal blood sample is taken to prepare a thin peripheral smear. The peripheral smear is dried, immersed in fixative, exposed to and incubated with an acid buffer, and stained with eosin. Under a microscope, the test reveals fetal cells to be stained a dark pink-red colour, whilst adult red cells appear pale or as uncoloured ghost outlines (Kim and Makar, 2011). Under a microscope, the fetal cells are counted and reported as a percentage of adult cells (Kim and Makar, 2011).
Flow cytometry using anti-fetal haemoglobin antibodies is a variant of flow cytometry that detects RhD positive fetal cells (Kim and Makar, 2011). In this method, monoclonal antibodies are directed against HbF (Davis, 2007). A maternal blood sample is collected and an RBC count is performed (Davis, 2007). Cells are then fixed and permeabilised with detergent to enable antibodies to enter the cellular membrane and bind HbF (Davis, 2007). A flow cytometer is used to analysed the antibody stained cells (Davis, 2007). This method uses positive and negative controls simultaneously to differentiation between fluorescence from fetal RBCs and non-specific background staining (Kim and Makar, 2011). The positive control is also highly important in setting out the parameters for gating a sample (Kim and Makar, 2011).
|
Rosette test |
Kleihauer-Betke acid elution test |
Flow cytometry using anti-fetal haemoglobin antibodies |
|
Advantages:
|
Advantages:
|
Advantages:
|
|
Disadvantages:
|
Disadvantages:
|
Disadvantages:
|
a) A >2mL bleed is detected via acid elution test in a sample taken from a D negative mother. Quantification via anti-D flow cytometry results in a zero bleed. Explain two possible reasons for the opposing results.
The differing results of the acid elution test and anti-D flow cytometry may be explained by the RhD status of the mother and fetus. If the mother is not RhD negative and is carrying an RhD positive fetus, this test would not reproduce the results of the acid elution test. Additionally, if the fetus has an RhD negative status, this would also cause the test to fail and detect a zero bleed. Furthermore, flow cytometry cannot accurately detect weak and partial D variants, resulting in a false negative result. Therefore, if the mother or fetus has either of these D variants, FMH would not be detected via flow cytometry. An alternative reason for the differing results between the two tests could be attributed to haemoglobinopathies, in which the flow cytometer detects HbF. Lastly, there could be a mistake in the tube, such that the antibody was not detected, causing an incorrect result.
b) Suggest a suitable test alternative to those already conducted to investigate the sample further and explain your reasoning for the alternative test.
The use of flow cytometry using anti-fetal haemoglobin antibodies would be a beneficial test to implement in order to clarify the results and detect if a bleed is present. This is important to ensure the appropriate and correct dose of RhIg is administered. Testing the sample using HbF flow cytometry would be useful as flow cytometry using anti-D failed to reproduce the results of the acid elution test.
There is a possibility that the acid-elution test produced a false positive result. The acid-elution test is limited in that is has poor accuracy and is prone to variations. The false positive may arise as a result of adult hereditary persistence of HbF, which is known in 1 to 2% of the population. Furthermore, during pregnancy the level of HbF rises by 25%. Therefore, the use of HbF flow cytometry would be able to detect if this is what caused the result.
The following bleeds were detected via anti-D flow cytometry in a D negative woman. Using the Mollison calculation work out:
- The bleed volume in mL
- The total RhIg dose in each case to the nearest 500 IU
- The top up RhIg dose required in each case to the nearest 500 IU
The Mollison calculation is used to calculate the volume of bleed. To do this, the background of the isotope matched control is subtracted from the number of events obtained in the D positive region, as recommended in the BCSH guidelines.
 The Mollison equation is as follows:
The Mollison equation is as follows:
D (+) events – D (-) events1800
FMH = ——————————— X ——-ml X 1.22
Total number of events 1
The equation can be simplified to the following:
FMH = % of D positive events x 18 x 1.22
-
- 0.81% x 18 x 1.22 = 17.79 mL
- 18 ml bleed* = 18 x 125 = 2250 IU – Rounded to nearest 500 = 2500 IU
- 2500 IU – 500 IU = 2000 IU
- 0.45 x 18 x 1.22 = 9.88 mL
- 10 mL bleed = 10 x 125 = 1250 IU – Rounded to nearest 500 = 1500 IU
- 1500 IU – 500 IU = 1000 IU
- 0.091 x 18 x 1.22 = 1.99 mL
- 2 mL bleed = 2 x 125 = 250 IU – Rounded to nearest 500 = 500 IU
- 500 IU – 500 IU = 0 IU
This bleed is under 4 mL and therefore no top up is required as 500 IU is routinely administered for a bleed of up to 4 mL.
-
- 1.09 x 18 x 1.22 = 23.94 mL
- 24 mL bleed = 24 x 125 = 3000 IU – Rounded to nearest 500 = 3500 IU
- 3500 IU – 500 IU = 3000 IU
- 0.02 x 18 x 1.2 = 0.43
- 1 mL bleed = 1 x 125 = 125 IU – Rounded to nearest 500 = 500 IU
- 500 IU – 500 IU = 0 IU
This bleed is under 4 mL and therefore no top up is required as 500 IU is routinely administered for a bleed of up to 4 mL.
References
Brinc, D. and Lazarus, A. (2009). Mechanisms of anti-D action in the prevention of hemolytic disease of the fetus and newborn. Hematology, [online] 2009(1), pp.185-191. Available at: http://asheducationbook.hematologylibrary.org/content/2009/1/185.long [Accessed 6 Mar. 2017].
de Haas, M., Thurik, F., Koelewijn, J. and van der Schoot, C. (2015). Haemolytic disease of the fetus and newborn. Vox Sanguinis, [online] 109(2), pp.99-113. Available at: https://www.ncbi.nlm.nih.gov/pubmed/25899660 [Accessed 6 Mar. 2017].
Dean, L. (2005). Blood groups and red cell antigens. 1st ed. [Bethesda, Md.]: NCBI.
Delaney, M. and Matthews, D. (2015). Hemolytic disease of the fetus and newborn: managing the mother, fetus, and newborn. Hematology, [online] 2015(1), pp.146-151. Available at: https://www.ncbi.nlm.nih.gov/pubmed/26637714 [Accessed 6 Mar. 2017].
Giancarlo Maria Liumbruno, Angelo D’Alessandro, Federica Rea, Vanessa Piccinini, Liviana Catalano, Gabriele Calizzani, Simonetta Pupella, Giuliano Grazzini (2010). Blood Transfus. 2010 Jan; 8(1): 8-16. doi: 10.2450/2009.0108-09
Kim, Y. and Makar, R. (2012). Detection of fetomaternal hemorrhage. American Journal of Hematology, [online] 87(4), pp.417-423. Available at: https://www.ncbi.nlm.nih.gov/pubmed/22231030 [Accessed 6 Mar. 2017].
Murray, N. and Roberts, I. (2007). Haemolytic disease of the newborn. Archives of Disease in Childhood – Fetal and Neonatal Edition, [online] 92(2), pp.F83-F88. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2675453/ [Accessed 6 Mar. 2017].
Roberts, I. (2008). The changing face of haemolytic disease of the newborn. Early Human Development, [online] 84(8), pp.515-523. Available at: https://www.ncbi.nlm.nih.gov/pubmed/18621490 [Accessed 6 Mar. 2017].
Urbaniak, S. and Greiss, M. (2000). RhD haemolytic disease of the fetus and the newborn. Blood Reviews, [online] 14(1), pp.44-61. Available at: https://www.ncbi.nlm.nih.gov/pubmed/10805260 [Accessed 6 Mar. 2017].
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allDMCA / Removal Request
If you are the original writer of this essay and no longer wish to have your work published on UKEssays.com then please click the following link to email our support team:
Request essay removal


