An Essay on Effective First Aid For Staphylococcus Aureus
| ✅ Paper Type: Free Essay | ✅ Subject: Biology |
| ✅ Wordcount: 2629 words | ✅ Published: 12 Jun 2018 |
The aim of this investigation is to experimentally determine which first aid product and its active ingredients are most effective against Staphylococcus aureus, and to establish how and why the chemical compounds of the active ingredients in each product affected the results.
Introduction
The idea for this exploration was developed as a result of an experiment and study that were conducted and taught in my medical microbiology class. The specific lesson that caught my interest was focused on the skin flora as well as infection causing bacteria. This, combined with the lab about the effects of specific antibiotics on bacteria as well as my considerable experience with first aid and disaster response skills led me to think about the importance of antibiotics for medication and treatments. I decided that my investigation would concentrate on topical first aid products against the occasionally pathogenic bacteria, Staphylococcus aureus, a member of the skin flora. My choice to pick a ubiquitous bacterium was because I wanted to focus on the more practical implementations of the investigation and could evaluate for myself which first aid product would be most useful in real world applications.
Background
This investigation requires background information about the biochemistry or mechanisms of action in specific compounds and the Kirby-Bauer test. These are described below.
Kirby-Bauer Test
The Kirby-Bauer test or disk diffusion tests allows for scientists to test the antibiotic sensitivity of bacteria. A disk is impregnated with a substance and placed on a petri dish and a zone of inhibition or inhibition zone appears after days or hours after incubation. The inhibition zone represents the area in which the bacteria has stopped growing or has been killed by the antibiotic. The size of the inhibition zone indicates the effectiveness of the antibiotic (the larger the diameter of the zone of inhibition the more effective the substance is).
Mechanism of Action in Compounds
This investigation focuses on five specific compounds which are active first aid antiseptic or antibiotic ingredients in the products that will be tested. These compounds are: benzalkonium chloride, triclosan, bacitracin zinc, polymyxin B sulfate, and neomycin sulfate. This information will be sectioned into Part A, B, C, D, and E.
Part A: Benzalkonium Chloride
Benzalkonium chloride is a member of the quaternary ammonium compounds (QACs) which are also known as cationic agents/surfactants. Furthermore, quaternary ammonium compounds have positively charged structures. Thus, the cationic zone of benzalkonium chloride disrupts the intermolecular attractions/electrostatic interactions of the negatively charged cell components, destroys the outer membrane, and ultimately kills the pathogen.
Part B: Triclosan
Triclosan works to inhibit bacterial growth through its mechanomolecular energy. This mechanomolecular energy is as a result of the ether single-bond rotations on the central oxygen atom. Subsequently, the rapid and fluctuating vibratory movements of the bonds in the molecule disrupt bacterial membranes which easily allow the chemical compound to enter the cell’s membrane. Once triclosan enters the cell membrane, it binds and blocks the active sites of the enoyl-acyl carrier-protein reductase enzyme (ENR) thus preventing the process of fatty acid synthesis. This fatty acid process is critical for building the pathogen’s cell membrane and its other vital functions necessary for processes like reproduction. Furthermore, at extremely low concentrations, triclosan can develop into a crystalline form by ring stacking, thus interfering with essential enzymes including the nicotinamide adenine dinucleotide (NADH) coenzyme of bacteria.
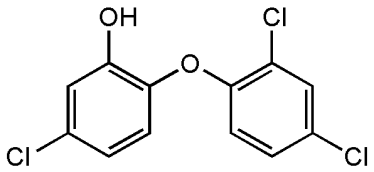
Figure 1: Figure 1 visualizes the two benzene rings and the central oxygen atom in the Triclosan compound.
Part C: Bacitracin Zinc
Bacitracin has antimicrobial activity primarily because of its ability to bind to divalent metal ions, in this case the Zn²º cation, resulting in bacitracin zinc. The Zn²º ion forms a ternary 1:1:1 antibiotic-metal-lipid complex. This means that the divalent metal ion complex can tightly bind to the lipid C‚…‚…-isoprenyl pyrophosphate molecules of the cell, acting like a bridge between the pyrophosphate and bacitracin zinc. Once the C‚…‚…-isoprenyl pyrophosphate has been compromised due to its inability to dephosphorylate or remove its phosphate (PO43ˆ’) through the pyrophosphatase enzyme and hydrolysis process, the pyrophosphate can no longer transport lipids into the cell-wall. Subsequently, this inhibits the process of cell-wall synthesis and results in the weakening of the cell wall and ultimately leads to bacterial death.
Part D: Polymyxin B Sulfate
The mechanism of action of polymyxin B sulfate is similar to that of benzalkonium chloride, in that it is also classified as a cationic surfactant. Therefore, like benzalkonium chloride, polymyxin B sulfate alters the external membrane of bacterial cells. Additionally, because of its positively charged amino group in the cyclic peptide region in the compound, it has an electrostatic attraction for the negatively charged lipopolysaccharide layer of bacterial cells and binds to these specific sites. Once these sites have been compromised, the outer membrane of the bacterial cell becomes destabilized and weakened.
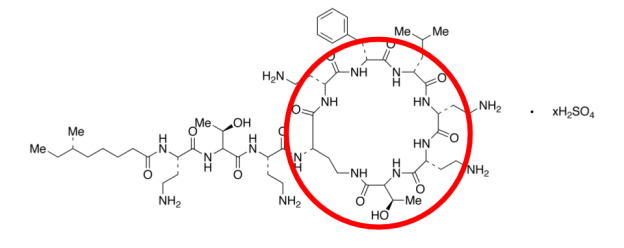
Figure 2: Figure 2 visualizes the amino group and cyclic peptide region of polymyxin B sulfate which is the primary mechanism of antimicrobial action within the compound.
Part E: Neomycin Sulfate
Neomycin sulfate is classified as an aminoglycoside antibiotic which means it has an amino group (-NH‚‚) attached to derivatives of sugar called glycosides. Aminoglycosides are highly positive in charge due to the presence of amino groups and have a high electrostatic attraction for the negatively charged outer surface of bacteria. This electrostatic interaction disrupts the membrane of the bacteria due to the displacement of Mg²º and Ca²º bridges and creates temporary openings in the bacterial cell membrane. Subsequently, this process causes intracellular content leakage and further increases the antibiotic intake in the bacteria. Additionally, rRNA molecules of bacteria are highly negative in charge because of the presence of phosphate groups. This negative charge has an electrostatic attraction with the positively charged antibiotic and allows the aminoglycoside to easily bind to the rRNA of the bacteria and thereafter inhibits the process of protein synthesis leading to bacterial cell death.
Prediction
The initial prediction is that NEOSPORIN® Original Ointment will be the most effective compared to Bactine Spray and CVS Health Instant First Aid Spray because it is a triple antibiotic and has a greater variety of active ingredients that can kill bacteria.
Experimental Procedure and Methodology
Variables
The independent variables for this experiment are the different first aid products because each product should influence the dependent variable (diameter of inhibition zone). The dependent variable is the size of the inhibition zones because the length depends on what first aid product is used. The controls of the investigation are the incubation time, petri dish, Staphylococcus aureus, method of inoculation, the incubator, and the amount of each drug because they are variables that are kept constant throughout each trial and for each product tested. It is important to maintain the controls throughout all trials so that measurements can be as consistent as possible.
Safety and Environmental Ethics
It is important to keep in mind the risks and safety precautions before attempting this experiment. These risks include residual bacterial contamination on the skin and burning. It is highly advised to wear gloves or rubber insulator gloves when needed and to wash hands frequently throughout the experiment. Additionally, an environmental ethical consideration must be taken in account because experimenting with antibacterial products can contribute to antibacterial resistance. However, the effects would be negligible due to the small scale size of the experiment.
Materials
- 125 ml of Agar
- Absorbent bibulous paper
- Hole puncher
- Sharpie
- Ruler (with millimeters)
- Gloves
- 12 Petri dishes
- Incubator @34 °C; set at 4.5
- 12 strips of Parafilm
- Forceps
- Tweezers
- Large beaker
- Hot plate
- Rubber insulator gloves
- Bunsen burner
- Strikers
- Inoculating loop
- Staphylococcus aureus
- NEOSPORIN® Original Ointment
- Bactine Spray
- CVS Health Instant First Aid Spray
Procedure
- Heat up 125 ml of agar in a beaker filled with water on a hotplate and wait until the agar is clear all the way through. Use forceps and rubber insulator gloves to remove the agar out of the beaker.
- Pour about the same amount of agar in each petri dish and wait until the agar sets (5-10 minutes).
- Use isolated Staphylococcus aureus and a heated and then cooled inoculating loop to carefully swab the culture into the 12 petri dishes. Make sure to go in a zigzag motion and cover all areas.
- Label each petri dish
- #1-#3: NEOSPORIN® Original Ointment
- #4-#6: Bactine Spray
- #7-#9: CVS Health Instant First Aid Spray
- #10-#12: Control
- Punch at least 12 holes into absorbent bibulous paper.
- Soak the disks with each of the drug solutions. Use tweezers that have been heated up and cooled down with water to minimize bacterial contamination. Additionally, each time a different product is being impregnated into the disks, reheat and cool the tweezers to minimize cross-product contamination.
- Place three disks containing NEOSPORIN® Original Ointment in petri dishes #1, #2, and #3.
- Place three disks containing Bactine Spray in petri dishes #4, #5, #6.
- Place three disks containing CVS Health Instant First Aid Spray in petri dishes #7, #8, #9.
- Leave the remaining three petri dishes with no disks in order to show that the petri dishes have pure cultures of Staphylococcus aureus.
- Use parafilm to seal all the petri dishes.
- Flip over all petri dishes and place in incubator at 34 °C on the 4.5 setting (the optimal temperature and conditions for bacterial growth)
- Measure the diameter of the inhibition zones (including the disk) 48 hours post inoculation with a ruler (in mm) and record data.
- Repeat all steps for Trial 2 and Trial 3 making sure all conditions are maintained.
Results
|
Product Name |
Table 1: Recorded Length of Inhibition Zones on Staphylococcus aureusᵃ |
|||
|
Trial 1 ± 0.5 mm |
Trial 2 ± 0.5 mm |
Trial 3 ± 0.5 mm |
Mean ± 0.5 mmᵇ |
|
|
NEOSPORIN® Original Ointment |
14.0 |
17.0 |
16.0 |
15.4 |
|
14.0 |
16.0 |
18.0 |
||
|
12.0 |
16.0 |
16.0 |
||
|
Bactine Spray |
12.0 |
16.0 |
18.0 |
16.3 |
|
16.0 |
17.0 |
18.0 |
||
|
16.0 |
14.0 |
20.0 |
||
|
CVS Health Instant First Aid Spray |
7.0 |
0.0 |
8.0 |
5.2 |
|
0.0 |
8.0 |
8.0 |
||
|
0.0 |
7.0 |
9.0 |
||
a- Diameter of zone of inhibition (mm) including disk diameter of 6mm
b- Average diameter of inhibition zone after 3 trials for each product
*The values that were found to have “0.0mm” were petri dishes that had no zone of inhibition
*Controls were not included as they only served to show that the petri dishes had pure samples of Staphylococcus aureus and are irrelevant to be included in the processed data
Conclusion and Evaluation
The objective of the experiment was to investigate the effectiveness of the active chemical ingredients in topical first aid treatments against Staphylococcus aureus. The initial aims of the investigation have been reached. The results of the experiment have suggested that Bactine Spray is the most effective against Staphylococcus aureus followed by NEOSPORIN® Original Ointment then CVS Health Instant First Aid Spray. Thus, the initial prediction was incorrect.
Bactine Spray contained 0.13% of benzalkonium chloride and in this investigation had an average length of 16.3 mm for its zone of inhibition (Table 1). According to a study done by Ali Fazlara (a member of the Department of Food Hygiene at Shahid Chamran University) and Maryam Ekhtelat (a researcher at Shahid Chamran University in the Department of Microbiology) found that because Staphylococcus aureus has a highly negative charge on its cell wall due to its slight anionic teichoic acids and peptidoglycan molecules, it allows for the cationic benzalkonium chloride to bind easily to specficically Staphylococcus aureus’ cell wall and thus block the active sites for essential enzymes to undergo their normal biochemical reactions for the bacterial cell. Therefore, benzalkonium chloride should theoretically be highly effective against Staphylococcus aureus. The data collected seems to support this and underpins the known fact that benzalkonium chloride is bacteriostatic (a chemical agent that stops bacteria from reproducing) at low concentrations. The investigation also suggests that NEOSPORIN® Original Ointment was the second most effective against Staphylococcus aureus compared with the Bactine Spray and CVS Health Instant First Aid Spray. The active ingredients within this first aid product are bacitracin zinc (400 units), neomycin sulfate (3.5mg), and polymyxin B sulfate (5,000 units). NEOSPORIN® Original Ointment resulted in an average zone of inhibition diameter of 15.4 mm, as shown in Table 1. The possible reason for why NEOSPORIN® Original Ointment was not as effective against Staphylococcus aureus in this investigation is because of antibacterial resistance to some or all of the active ingredients by the bacteria. Studies have proposed that the isolate primarily found in the United States called USA300 methicillin-resistant Staphylococcus aureus (MRSA), has been increasingly discovered to have been resistant to neomycin sulfate, bacitracin zinc, and polymyxin B sulfate. In this investigation, it can be suggested that CVS Health Instant First Aid Spray, with a concentration of 0.13% of triclosan was the least effective against Staphylococcus aureus. It resulted in an average zone of inhibition diameter of 5.2 mm according to Table 1. A possible conclusion that can be reached based on the results is the increasingly proven theory that Staphylococcus aureus has a progressively high antimicrobial resistance to triclosan. This is due in part because triclosan was the most common active ingredient in over the counter (OTC) products, which contributed greatly to Staphylococcus aureus’ antimicrobial resistance. As a matter of fact, the FDA banned triclosan on consumer antibacterial wash products because of the health related risks from bacterial resistance. However, some products still use triclosan because it banned to be used in soaps.
These conclusions are incomplete and require improvements in order to thoroughly and further confirm the results and achieve consistent values. The addition of more trials would further eliminate any systematic errors that may have occurred such as error when impregnating disks with the products or cross-product contamination. Any instances of random error can be best alleviated by the use of a Vernier calliper (a measuring instrument that is used for measuring diameters) instead of a ruler. The use of a calliper to measure would contribute to higher precision and less measurement uncertainty.
Extending the investigation to other normal bacterial skin flora would be interesting to see the extent of the effectiveness of Bactine Spray, NEOSPORIN® Original Ointment, and CVS Health Instant First Aid Spray on different bacteria.
Works Cited
Chittapragada, Maruthi, and Sarah Roberts. Aminoglycosides: Molecular Insights on the Recognition of RNA and Aminoglycoside Mimics. Perspectives in Medicinal Chemistry, 2009. www.ncbi.nlm.nih.gov/pmc/articles/PMC2754922/. Accessed 15 Feb. 2017.
Economou, Nicoleta J., et al. High-resolution crystal structure reveals molecular details of target recognition by bacitracin. 2013. www.pnas.org/content/110/35/14207.full.pdf. Accessed 13 Feb. 2017.
Fazlara, Ali, and Maryam Ekhtelat. The Disinfectant Effects of Benzalkonium Chloride on Some Important Foodborne Pathogens. IDOSI, 2012. www.idosi.org/aejaes/jaes12(1)12/4.pdf. Accessed 16 Feb. 2017.
Federal Drug Administration. “FDA Issues Final Rule on Safety and Effectiveness of Antibacterial Soaps.” U S Food and Drug Administration Home Page, 2 Sept. 2016, www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm517478.htm. Accessed 16 Feb. 2017.
Kaya, Deniz. “Quarternary Ammonium Compounds.” 21 Jan. 2010, Accessed 9 Feb. 2017.
Kling, Jim. “Antibiotic Ointments May Fuel Resistance and Spread of MRSA.” Medscape, 14 Sept. 2011, www.medscape.com/viewarticle/749666. Accessed 16 Feb. 2017.
Maxka, Jim. “Organic Chemistry Interactive Notes.” Organic Chemistry, North Arizona University. Arizona. Reading.
McDonnell, Gerald, and A. D. Russell. Antiseptics and Disinfectants: Activity, Action, and Resistance. Clinical Microbiology Reviews, 1999. www.ncbi.nlm.nih.gov/pmc/articles/PMC88911/. Accessed 9 Feb. 2017.
Petersen, Richard C. Triclosan Antimicrobial Polymers. HHS Public Access, 2016. www.ncbi.nlm.nih.gov/pmc/articles/PMC4893770/. Accessed 12 Feb. 2017.
—. Triclosan Computational Conformational Chemistry Analysis ForAntimicrobial Properties in Polymers. HHS Public Access, 2015. www.ncbi.nlm.nih.gov/pmc/articles/PMC4394635/. Accessed 12 Feb. 2017.
“Polymxyin B Sulfate.” Digital Photograph. Accessed 13 Feb. 2017.
Pub Chem. “Aerosporin | C56H100N16O17S – PubChem.” The PubChem Project, pubchem.ncbi.nlm.nih.gov/compound/Polymyxin_B_sulfate#section=Related-Compounds. Accessed 13 Feb. 2017.
Ramin Khajavi, Morteza Sattari and Ali Ashjaran, 2007. The Antimicrobial Effect of Benzalkonium Chloride on Some Pathogenic Microbes Observed on Fibers of Acrylic Carpet. Pakistan Journal of Biological Sciences, 10: 598-601.
Stone, K. J., and Jack L. Strominger. Mechanism of Action of Bacitracin: Complexation with Metal Ion and C55-Isoprenyl Pyrophosphate. 1971. www.ncbi.nlm.nih.gov/pmc/articles/PMC389626/pdf/pnas00087-0326.pdf. Accessed 12 Feb. 2017.
Tay, William M., et al. 1H NMR, Mechanism, and Mononuclear Oxidative Activity ofthe Antibiotic Metallopeptide Bacitracin: The Role of D-Glu-4,Interaction with Pyrophosphate Moiety, DNA Binding andCleavage, and Bioactivity. JACS Articles, 2010. Accessed 13 Feb. 2017.
Unblok Bio Solutions. “Ammonium.” Unblok Bio-Fix, unblok.co/ammonium/. Accessed 9 Feb. 2017.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allDMCA / Removal Request
If you are the original writer of this essay and no longer wish to have your work published on UKEssays.com then please click the following link to email our support team:
Request essay removal


