Physicochemical Events in Production of Butter and Margarine
| ✅ Paper Type: Free Essay | ✅ Subject: Sciences |
| ✅ Wordcount: 2520 words | ✅ Published: 11 Apr 2018 |
- Byeong-Keon LEE
Introduction
Margarine and butter are known world-widely as spread, and have dominated the production in the western world, such as America including New Zealand. Recently, Asian countries particularly China and India have shown an increase of butter consumption. The amount of butter China consumes was predicted to increase to 13% over the next decade (Fallow, 2013). Both margarine and butter have similar a taste, texture as well as nutritional values. The characteristic of both products are significantly similar – for example, water-in-emulsion and the fat content are approximately in 81% range. However, the two products are actually different when it is compared with the ingredients and the structure. The argument surrounding the diversity of butter and margarine is typically concerned with human health, the production of margarine is a man-made food which follows a lot of chemical treatments and speculation that it might cause several diseases (Guyenet, 2008). On the other hand, the butter contains a higher level of cholesterol that can negatively influence human digestion (Collins, 2013). In this assignment, physical and chemical properties and psychochemical processes and the changes of making margarine and butter will be discussed.
Physical and Chemical difference
Both butter and margarine are triglyceride (triacylglycerol) in an ester linkage composure of glycerol and 3 fatty acids that also uses the water-in-oil emulsion process where water is dispersed in phase and oil in a continuous phase. The critical comparison and contrast between butter and margarine will be the ingredients which have different states in room temperature and the chemical process of making it.


Butter is an animal dairy product which is composed of a complex chain of saturated fat and unsaturated fatty acid, and a high concentration of animal cholesterol. As it’s clearly seen from the picture, the majority of fatty acids are saturated and some fatty acid unsaturated. The color of butter is normally yellow indicating it contains small amounts of carotene another meaning of vitamin A. (Reyes, n.d). The higher saturation levels of the fatty acids, contributes to the physical structure of the animal fat. Higher saturation levels are highly correlated with higher London Dispersion forces. Hence, due to the high London Dispersion Forces holding the saturated bonds together, animal fat is solid at room temperature (Brown, LeMay and Bursten, n.d.).

Figure 2. Percentage composition of fatty acid in butter

 In contrast, margarine is a substitution of butter spread made from vegetable oil. Oil has a lot of double bonds, which typically disrupts the London dispersion forces between fatty acids and low effective surface area which the molecules do not pack together giving a lower melting point
In contrast, margarine is a substitution of butter spread made from vegetable oil. Oil has a lot of double bonds, which typically disrupts the London dispersion forces between fatty acids and low effective surface area which the molecules do not pack together giving a lower melting point
The mixed vegetable oil, like soyabean oil or corn oil has a structure of mono-unsaturated or polyunsaturate of fatty acids and those unsaturated fats that need to be converted into a semi-solid to make margarine. This process is called hydrogenation or additional reaction, by adding hydrogen gas with nickel catalyst in a double bond of unsaturated fat. The double bond become a single bond, and this makes the molecules pack together better. In other words, the London dispersion forces become active and the interaction between the molecules is called the Van Der Waals interaction (Burrows, 2009). The results of the hydrogenated vegetable oil, are of it process to solidify at room temperature. The majority of fatty acid in margarines are unsaturated. Most fatty acids have a cis-form and margarine is unlikely to form trans-fatty acid. During the hydrogenation, some of the fatty acid will tend to form trans-addition.

Figure 4. Hydrogenation reaction occur in unsaturation reaction
Chemical process of butter
The milk itself come from the cow which does not have to add artificial ingredients. Therefore butter contains nutritional values such as fat, protein and different types of vitamins which are found originally in milk. The chemical process is outlined in figure 4, that depicts the chemical process. For example pasteurization – the ripening, aging and churning are the most significant factors for making butter cream.
 1) Milk separation
1) Milk separation
 The standard raw milk mostly contains 15 to 25 percent of fat globules, the globule is a tiny membrane filled with the fat molecule (Murphy, 2011). When the raw milk is shaken, the globules’ membranes will crash against each other and break. As regard, the fat will start to burst out and tend to bunch together with the contents of other burst globules, consequently, the butter cream is separated from raw skim milk and the butter cream will contain approximately 38% of fat.
The standard raw milk mostly contains 15 to 25 percent of fat globules, the globule is a tiny membrane filled with the fat molecule (Murphy, 2011). When the raw milk is shaken, the globules’ membranes will crash against each other and break. As regard, the fat will start to burst out and tend to bunch together with the contents of other burst globules, consequently, the butter cream is separated from raw skim milk and the butter cream will contain approximately 38% of fat.
2) Pasteurization
 The raw cream has to be pasteurized to a temperature of 95oC for 15 seconds to kill any interference in production of butter particularly enzyme and micro-organisms. This process is widely known as HTST (High Temperature Short Time) pasteurization. It is very interesting to note that nowadays, dairy industries implements UHT (Ultra High Temperature) pasteurization procedure to produce lower number of micro-organism, which leads to longer shelf life. HTST process are more favorable compared to UHT as in the production of butter, Lactobacillus sp. is wanted to further ferment the cream, giving of specific flavors. In contrast, if the cream is subjected to UHT, any bacteria present in the cream would be eliminated, further contributing to no flavor compound formation during the ripening process (Butler and Media, n.d).
The raw cream has to be pasteurized to a temperature of 95oC for 15 seconds to kill any interference in production of butter particularly enzyme and micro-organisms. This process is widely known as HTST (High Temperature Short Time) pasteurization. It is very interesting to note that nowadays, dairy industries implements UHT (Ultra High Temperature) pasteurization procedure to produce lower number of micro-organism, which leads to longer shelf life. HTST process are more favorable compared to UHT as in the production of butter, Lactobacillus sp. is wanted to further ferment the cream, giving of specific flavors. In contrast, if the cream is subjected to UHT, any bacteria present in the cream would be eliminated, further contributing to no flavor compound formation during the ripening process (Butler and Media, n.d).

3) Inoculation and ripening
 After pasteurization, then for some time the helpful bacteria ripens in the raw cream so it can proliferate to render it in a better condition for butter making. The butter maker has made significant improvements through experience, that ripened cream churns more rapidly than sweet cream. It take about 24 to 48 hours for the cream to fully ripen (Marina. 2011).
After pasteurization, then for some time the helpful bacteria ripens in the raw cream so it can proliferate to render it in a better condition for butter making. The butter maker has made significant improvements through experience, that ripened cream churns more rapidly than sweet cream. It take about 24 to 48 hours for the cream to fully ripen (Marina. 2011).
 The lactic acid bacteria (Lactobacillus helveticus) will actually further ferment or ripening the cream and breaking down the long fatty acid into short fatty acid (diacetyl compounds). This short fatty acid has which has unique taste and good smell. An increase in lactic acid causes a reduction in overall pH which cause the proteins in milk to change. As cream returns to room temperature, the lactose in milk begins to ferment and creating lactic acid which help to make butter. (Belitz, Grosch and Schieberle, 2009)
The lactic acid bacteria (Lactobacillus helveticus) will actually further ferment or ripening the cream and breaking down the long fatty acid into short fatty acid (diacetyl compounds). This short fatty acid has which has unique taste and good smell. An increase in lactic acid causes a reduction in overall pH which cause the proteins in milk to change. As cream returns to room temperature, the lactose in milk begins to ferment and creating lactic acid which help to make butter. (Belitz, Grosch and Schieberle, 2009)
4) Agitation
It is a physical change which the process of churning butter consists of the mixing that separates some of the fatty solids out of milk. The formation of fat crystal can be influence by suitable temperature control during the ripening process. Fat molecule in cream are surrounded by membranes made of phospholipids and proteins which prevent the fat from pooling together. Fat globules are bumping into each other and membrane is breaking down when it happens the fat globules are trying to stick together. Agitation cream damage these membrane allowing the fats to pool together and form whipping cream.

5) Churning
 Churning is essentially strong mechanical cream shearing which tears the membranes of the fat globules and facilitates coalescence of the globules. The cream “breaks” any tiny granules of butter appear. In the churning compartment, a rotating impact wave cause butter granule formation. The separation compartment is divided into two parts. The butter is first churned further, resulting in the formation of butter granules of larger diameter. As the churning continues fat molecule become popcorn butter which separate from butter milk. The popcorn butter has the same consistency as regular butter at room temperature. They put the butter cream on bulk tank where mixers sterile to maintain the consistency. If agitation and churning of the cream continues, finally a solid is formed(butter-cream) and the remaining liquid (butter milk) is separated (Ripema, 1970).
Churning is essentially strong mechanical cream shearing which tears the membranes of the fat globules and facilitates coalescence of the globules. The cream “breaks” any tiny granules of butter appear. In the churning compartment, a rotating impact wave cause butter granule formation. The separation compartment is divided into two parts. The butter is first churned further, resulting in the formation of butter granules of larger diameter. As the churning continues fat molecule become popcorn butter which separate from butter milk. The popcorn butter has the same consistency as regular butter at room temperature. They put the butter cream on bulk tank where mixers sterile to maintain the consistency. If agitation and churning of the cream continues, finally a solid is formed(butter-cream) and the remaining liquid (butter milk) is separated (Ripema, 1970).
6) Washing, Cooling, and Vacuum
Subsequently, the buttermilk is separated and the butter is washed if necessary. which cause the butter-cream fat molecules to bunch together releasing water and air. In addition, cooling process and vacuuming is performed to reduced to water and air content to 1% (Belitz, Grosch and Schieberle, 2009).
Chemical process of margarine
 Most margarines are made from a variety substances such as vegetable oil and edible animal fats. It composed of approximately a 80% combination of fat, that is either saturated fat or unsaturated fats, approximately 18% of liquid which derives from either pasteurized skim milk or a soybean protein fluid (Ripema, 1970).
Most margarines are made from a variety substances such as vegetable oil and edible animal fats. It composed of approximately a 80% combination of fat, that is either saturated fat or unsaturated fats, approximately 18% of liquid which derives from either pasteurized skim milk or a soybean protein fluid (Ripema, 1970).
1) Crude oil
 Different types of vegetable oil like corn and soybean are chemically extracted and refined to form crude oil and the crude oil can be neutralized and treated with a caustic soda solution that removes free fatty acid, and corrosive and pungent sulphur compounds (Sample, 2009). The free fatty acid, influences an undesirable taste. The process follows by washing then mixing with water, so it can separate, it is then left to dry with the aid of vacuum.
Different types of vegetable oil like corn and soybean are chemically extracted and refined to form crude oil and the crude oil can be neutralized and treated with a caustic soda solution that removes free fatty acid, and corrosive and pungent sulphur compounds (Sample, 2009). The free fatty acid, influences an undesirable taste. The process follows by washing then mixing with water, so it can separate, it is then left to dry with the aid of vacuum.
2) Modification
Margarine is not originally a yellow colour which the decolourisation process is for removing the grey colour by the absorption to activated clay or bleaching process (Ripema, 1970). The bleaching process utilizes bleaching earth and charcoal material in the vacuum condition. The process helps to absorb any undesirable colorants, which will be later filtered out from the oil mixture. Other sustenance impurities are both organic and inorganic, which naturally occurs raw oil that is removed by the de-acidification process using akali treatments.
3) Hydrogenation
 The hydrogenation is used as catalyst called nickel and the structure varies by cis-addition and trans-addition. This process helps the animal and vegetable oils blend and are able to let the formation from a liquid into a fat that is in a state that increases the melting point. However only some of the double bonds of polyunsaturated vegetable oil are hydrogenated and named partial hydrogenation. Therefore the degree of hydrogenation can be varied to give softer or harder margarine. (Haynes, 2014)
The hydrogenation is used as catalyst called nickel and the structure varies by cis-addition and trans-addition. This process helps the animal and vegetable oils blend and are able to let the formation from a liquid into a fat that is in a state that increases the melting point. However only some of the double bonds of polyunsaturated vegetable oil are hydrogenated and named partial hydrogenation. Therefore the degree of hydrogenation can be varied to give softer or harder margarine. (Haynes, 2014)
Figure 10. Chemical reaction of hydrogenation
4) Deodorization
 The modification is complete so the base oil is prepared to reproduce similar qualities to butter, this stage decrease unwanted smells and taste (Formo, n.d). The removal of the poor smell is by a batch process that steams the oil, the temperature used is very high and reaches from175–205°C in Europe and 235°–250° C in America, during this process the bad tastes and smells are taken away by the extractor fan. The oil can then be either further processed to make margarine or alternatively vegetable oil. (Singh, P. n.d.)
The modification is complete so the base oil is prepared to reproduce similar qualities to butter, this stage decrease unwanted smells and taste (Formo, n.d). The removal of the poor smell is by a batch process that steams the oil, the temperature used is very high and reaches from175–205°C in Europe and 235°–250° C in America, during this process the bad tastes and smells are taken away by the extractor fan. The oil can then be either further processed to make margarine or alternatively vegetable oil. (Singh, P. n.d.)
5) Emulsify
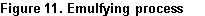 When the emulsion of the stabilized immiscible liquids is blended with the use of an emulsifier. The way margarine is constructed is through the emulsion process that adds salty water droplets in oil, as well as an emulsion ingredient to a cup. Salt content is modified as an emulsifier and renamed lecithin, the final stage is the separating of starch and emulsifier that forms a consistent mixture. Moreover, all the nutrients and color in the margarine are artificially made particularly vitaminisation processed into the margarine, to have a measured amount of vitamin with butter (Hasenhuettl and Hartel, 2008).
When the emulsion of the stabilized immiscible liquids is blended with the use of an emulsifier. The way margarine is constructed is through the emulsion process that adds salty water droplets in oil, as well as an emulsion ingredient to a cup. Salt content is modified as an emulsifier and renamed lecithin, the final stage is the separating of starch and emulsifier that forms a consistent mixture. Moreover, all the nutrients and color in the margarine are artificially made particularly vitaminisation processed into the margarine, to have a measured amount of vitamin with butter (Hasenhuettl and Hartel, 2008).
6) Cooling and kneading process
The method for the cooling an kneading process has two ways of completing, either with a tube chiller or a chilling drum-complector. Tube chiller method uses a closed system that has a one step process. Unlike the Chilling Drum-Complector that has a longer process, allowing cooling and kneading that lets the mixture rest, and the crystallization of fats at slower pace. However the advantage of the tube chiller is the reduced capacity of spoilage compact size in relation to production levels, and ease of operation. (Hasenhuettl and Hartel, 2008).
Conclusion
In conclusion, butter and margarine have significantly different physical and chemical properties as well as physiochemical duties in the production. Although the nutrition and texture of products are the same, but the ingredients of butter and margarine are different and this leads to a structure of both products that have different qualities. Moreover, both products have a different chemical process. In fact, margarine has extra steps to convert the butter from a liquid state to a solid state, that furthers the need to process, using hydrogenation.
References
Reyes, V. (2013). Chemical structure of butter. [Figure 1] Available at: http://scienceandfooducla.wordpress.com/2013/05/28/homemade-butter/ [Accessed 15 May. 2014].
P. B. Hawk, O. Bergeim, Blakiston, (2010). Fatty acids in butter. Percentage composition from Practical Physiological Chemistry,. [Figure 2] Available at: http://antoine.frostburg.edu/chem/senese/101/consumer/faq/butter-composition.shtml [Accessed 15 May. 2014].
Image by user called Smokefoot, (2012). Hydrogenation of fatty acid. [Figure 3] Available at: http://en.wikipedia.org/wiki/File:H2forMargerin.png#filehistory [Accessed 15 May. 2014].
Alpha Omega trial, (n.d.). Margarine composition in percentage. [Figure 4] Available at: http://en.wikipedia.org/wiki/File:H2forMargerin.png#filehistory [Accessed 15 May. 2014].
Yokogawa Corporation, (2007). Stable and Accurate Density Measurement to Control Fat Content in Skim Milk. [figure 5] Available at: http://www.yokogawa.com/us/technical-library/application-notes/stable-and-accurate-density-measurement-to-control-fat-content-in-skim-milk.htm [Accessed 15 May. 2014].
Andrews, R. (2012). All About Milk. [Figure 6] Available at: http://www.precisionnutrition.com/all-about-milk [Accessed 15 May. 2014].
Shannon, (2011). Making A Big Batch of Cultured Butter. [Figure 8] Available at: http://www.nourishingdays.com/2011/10/making-a-big-batch-of-cultured-butter/ [Accessed 15 May. 2014].
Castelli, C. (2008). Crude Corn Oil. [Figure 9] Available at: http://www.alibaba.com/product-detail/Crude-Corn-Oil_144278983.html [Accessed 15 May. 2014].
Quadro Engineering, (n.d.). Production of Margarine and Low Fat Spreads. [Figure 11] Available at: http://www.quadroytron.com/menu_app/food_margarine.asp [Accessed 15 May. 2014].
Belitz, H., Grosch, W. and Schieberle, P. (2009). Food chemistry 4th revised and extended edition. 4th ed. Springer, pp.pg 526 – 527.
Collins, C. (2013). What’s healthier, butter or margarine?. NZ Hearald. [online] Available at: http://www.nzherald.co.nz/lifestyle/news/article.cfm?c_id=6&objectid=11169963 [Accessed 15 May. 2014].
Fallow, B. (2013). China’s slowdown may be good for NZ. NZ Hearld. [online] Available at: http://www.nzherald.co.nz/business/news/article.cfm?c_id=3&objectid=10902631 [Accessed 15 May. 2014].
Formo, M. (n.d.). fat and oil processing (chemistry) :: Deodorization. [online] Encyclopedia Britannica. Available at: http://www.britannica.com/EBchecked/topic/202405/fat-and-oil processing/50167/Deodorization [Accessed 15 May. 2014].
Guyenet, S. (2008). Whole Health Source: Butter, Margarine and Heart Disease. [online] Wholehealthsource.blogspot.co.nz. Available at: http://wholehealthsource.blogspot.co.nz/2008/12/butter-margarine-and-heart-disease.html [Accessed 15 May. 2014].
Hasenhuettl, G. and Hartel, R. (2008). Food emulsifiers and their applications. 1st ed. New York: Springer, p.271.
Haynes, F. (2014). Take the Guess Work from Figuring Out the Trans Fats in Your Foods. [online] About.com Low Fat Cooking. Available at: http://lowfatcooking.about.com/od/faqs/f/hydrogenated.htm [Accessed 15 May. 2014].
Marina, (2011). Cultured butter – CULTURED, AGED, BREWED. [online] Culturedagedbrewed.com. Available at: http://www.culturedagedbrewed.com/2011/12/20/cultured-butter/ [Accessed 15 May. 2014].
Murphy, L. (2011). Emulsion Explosion: How to Make Butter. [online] Scientificamerican.com. Available at: http://www.scientificamerican.com/article/bring-science-home-butter-emulsion/ [Accessed 15 May. 2014].
Singh, P. (n.d.). fat and oil processing (chemistry) :: Deodorization. [online] Encyclopedia Britannica. Available at: http://www.britannica.com/EBchecked/topic/202405/fat-and-oil processing/50167/Deodorization [Accessed 15 May. 2014].
Ripema, S. (1970). The Story of Margarine. 1st ed. Public Affairs Press.
Sample, I. (2009). Trafigura case: toxic slop left behind by caustic washing. [online] the Guardian. Available at: http://www.theguardian.com/environment/2009/sep/16/trafigura-case-toxic-slop [Accessed 15 May. 2014].
Butler, C. and Media, D. (n.d.). Why Can’t You Use Ultra Pasteurized Cream for Making Butter?. [online] Everyday Life – Global Post. Available at: http://everydaylife.globalpost.com/cant-use-ultra-pasteurized-cream-making-butter-42002.html [Accessed 15 May. 2014].
Burrows, A. (2009). Chemistry3. 1st ed. Oxford: Oxford University Press, pp. page 85 to page 86.
Brown, L., LeMay, E. and Bursten, B. (n.d.). Chapter 11 intermolecular Forces, Liquids and Solids. 10th ed. [ebook] The central science, pp.slide 18 to slide 23. Available at: http://alpha.chem.umb.edu/chemistry/ch115/Mridula/CHEM 116/documents/chapter_11au.pdf [Accessed 15 May. 2014].
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allDMCA / Removal Request
If you are the original writer of this essay and no longer wish to have your work published on UKEssays.com then please click the following link to email our support team:
Request essay removal


