Characteristics and Properties of Titanium dioxide
| ✅ Paper Type: Free Essay | ✅ Subject: Sciences |
| ✅ Wordcount: 1933 words | ✅ Published: 15 Sep 2017 |
MANOJNA EDARA

TITANIUM DIOXIDE
History:
Titanium dioxide Characterization:
Bright unmistakable ideology and the scope of titanium dioxide nanoparticles are as follows. The UV-Visible maintenance scope of titanium dioxide nanoparticles was sifted in the region of 200 and 700nm. The ingestion of this is most extraordinary occurs at 340nm and this is the thing that it exhibits a band opening estimation of 3.6 eV.DRS ultra violet-Visible spectrum. The DRS-UV-Visible supernatural examination of titanium dioxide nanoparticles was done in the region of in the middle of 200 and 700nm.The plot of absorbance verses wave length is can be watched. The range portrays increase in absorbance just underneath 400nm and the reason is of band hole excitation. By taking 350nm as the onset of absorbance the band gap regard was figured to be 3.5eV. The band gap regard is dependable with that of Ultra Violet Visible ingestion scope of titanium dioxide nanoparticles. Sharp absorbance lines underneath 250nm are the reason due to this titanium dioxide nanoparticles and of size generously very littler than those fascinating around 350nm.

This is the image of amorphous titanium dioxide.
Structure and synthesis:
For a physical approach, the metallic nanoparticles can be generally fused by evaporation condensation, which could be finished by using a tube warmer at barometrical weight. In any case, by virtue of using a tube warmer at air weight there are a couple burdens, for instance, a gigantic space of tube radiator, unfathomable use imperativeness for raising the regular temperature around the source material and an impressive measure of time for fulfilling warm robustness. In this way, unique techniques for mix of Ag nanoparticles in perspective of the physical approach have been delivered and in the combination of titanium dioxide nanoparticles, Titanium tetra isopropoxide was utilized as an antecedent and was blended with hcl, ethanol and deionized water mixture, stirred for 60 minutes, which pH scope of 1.5.Amount of around 10ml of deionized water was added to the above blend and it was mixed for almost 2 hours at room temperature and at last the arrangement was dried at temperature and the powder was warmed at 120 degree for 60 minutes and the crystalline character of titanium dioxide was checked by electron diffraction. The electron diffraction configuration exhibits that the cases are made out of an a crystalline shape. Because of the example tio2: Ag diffuse rings in diffraction illustration are evident The diffraction case of the crystalline example tio2:Ag0 certifies its anatase outline .Results procured by electron diffraction relate with the Xray diffraction comes to fruition and the x beam diffraction of the shapeless and crystalline titanium dioxide doped with silver (tio2:Ag0 and TiO2a:Ag0) a. All appearance in the diffraction cases of titanium dioxide were recognized as they are having a place with anatase and in the case of the vague titanium dioxide, no reflections are viewed. In any case, practically identical as in, there are no obvious apexes exhibiting the closeness of silver in the xray diffraction of the silver doped titania tests. It probably comes to fruition due to minimal size of silver nanoparticles which are under 5nm and its low substance which is underneath 2 percent and conversely with Chao et al. titanium dioxide powder calcined starting at now at 400°C is especially cemented. The mass touchy X-beam diffraction examples were made to brought with Philips XPer diffractometer at a room temperature utilizing monochromatic excitation and the estimations were taken under pillar increasing speed conditions and even though the surface delicate X-beam photoelectron spectroscopy estimations were performed under ultra-high vacuum condition and in a frame work that shows a base weight. Keeping in mind the final result to concentrate the synthetic condition of titanium and oxygen in the nanoparticles we utilized a standard non-monochromatized X-beam source and a VG Clam 4 electron spectrometer. The spectra were redressed for X-beam satellites and optional electron foundation preceding examination. Filtering electron microscopy pictures of similar examples were recorded with a LED magnifying lens utilizing an electron bar vitality of 15 keV and a shaft current of 2.62 A.
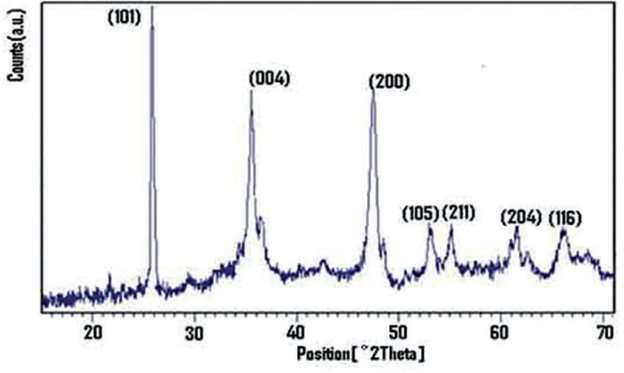
This is the x- ray diffraction pattern of titanium dioxide nanoparticles.
Properties:
The physicochemical properties of nanoparticles decide both their destiny in the earth and their valuable and destructive impacts. Despite the fact that the span of the nanoparticles can be the most recognizing property when contrasted with ordinary particles, other special physical and compound properties start to rise as particles approach the nanoscale extend. Therefore, Auffan propose that under a basic size, it is unrealistic to just scale the properties of mass materials in view of the surface range to anticipate the properties of nanoparticles”. Different researchers concur that “albeit some material properties, similar to substance creation and gem structure, are the same on the nanoscale as in the mass stage, different properties vary … a nanoparticle holds properties of both materials in the mass stage and sub-atomic forerunners”. Undoubtedly a few reviews bolster these affirmations and in any case, different analysts have found that inspite of the way that the physicochemical properties contrast, the impacts of nano silver can be like those delivered by traditional ionic silver. These varying discoveries drove the Federal Insecticide, Fungicide, and Rodenticide Act Scientific Advisory Panel to infer that comparison of physicochemical properties of the nano mass materials are required. Exactly which physiochemical properties of designed nanoparticles, including nano Ag, can be helpful for foreseeing their conduct and collaborations in nature is indistinct. A few associations and autonomous scientists have distributed proposals on the physicochemical portrayal information that ought to go with research discoveries on transport, change, and destiny forms and biological and human poisonous quality. These suggestions depend on a combination of distributed, companion surveyed ponders on the conduct and impacts of nanoparticles, however the prescribed properties fluctuate by association and scientist. A portion of the suggestions with respect to portrayal some time recently, amid, and after danger studies are further depicted in Section 6.1.1. All in all, the most endorsed physicochemical properties include:
• Size, including bunching inclinations.
• Morphology, including shape and precious stone structure.
• Surface range;
• Chemical piece;
• Surface science and reactivity;
• Solubility; and
• Conductive, attractive, and optical properties.
Advantages:
The ability to rot a broad assortment of possible characteristic toxic substances and NOx gasses consolidated with an especially adaptable era handle that engages the control over titanium dioxide nanoparticle pearl structure, atom size and crystallinity which gives the variable courses of action as tailorâ€made materials for a given issue or application our customers may experience thus far our titanium dioxide photograph impetuses have been adequately striven for the going with applications: these are for various improvement materials like strong, strong tiles, stoneware tiles, housetop tiles, as an additional substance or as a thin layer. The last improvement materials show a high photocatalytic affect, which enables normal toxin departure and NOx gas removal for various responses for the remediation of polluted air made by warm power stations and significant movement for remediation of mechanical waste water defiled by specific common fragments seeks hydrophilic and photo catalytically dynamic thin layers on various substrates like glass easy-to-clean surfaces.
Titanium dioxide in arrangement or suspension can be utilized to sever protein that contains the amino corrosive proline at the site where proline is available. Because of the critical ionic and electronic conduction of Titanium dioxide, it is intense to be utilized as the blended conductor.
Disadvantages:
This can experience a substance response in sunlight and research uncovered that titanium dioxide got from rutile which is a reddish brown colored mineral which had titanium dioxide and was less hurtful than titanium dioxide got from anatase which is used as pigments in paints, the same as covered titanium dioxide. Titanium dioxide is to a great degree viable in reflecting light, making it a breathtaking white color. This is normally extremely helpful on the off chance that you need to make a splendid white toothpaste. Having reflecting properties its great light make it viable as a sun filter. The blend of daylight, water and titanium in nano-frame makes hydroxyl radicals and these assault the covering of the rooftop. This outcomes in quickly maturing rooftops which ought to keep going for a considerable length of time.
Uses:
The capacity to break down an extensive variety of conceivable natural poisons and NOx gasses combined with an exceedingly adaptable creation which prepares that empowers the control over Titanium dioxide nanoparticle precious stone structure, molecule size and crystallinity that gives the variable arrangements as tailorâ€made materials for a given issue or application our clients may experience. Titanium dioxide photo catalysts have been effectively tried for the accompanying applications:
For different development materials such as solid tiles, earthenware tiles, rooftop tiles in the type of an added substance or as a thin layer. The last development materials display like a high photocatalytic impact, which empowers natural poison expulsion and NOx gas evacuation
- for different answers for the remediation of dirtied air created by warm power stations and unexpected overwhelming movement .
- for treatment of modern waste water which was contaminated by particular natural segments
- for hydrophilic and photo catalytically dynamic thin layers on different substrates which were easy to clean like glass.

This figure(a) shows the photo catalysts for air remediation by adding titanium dioxide nanoparticles in roof or concrete tiles.
Environmental impacts:
It was affirmed that the assessed titanium dioxide nanoparticles, utilized at a focus up to 25% as an UV channel in sunscreens, can be viewed as safe for people after application on a solid, in place or sunburnt skin. Marking of the restorative items may not demonstrate this focus on the bundle, but the security appraisal depends on the current logical information, which demonstrates that neither titanium dioxide in Nano frame nor in non-nano shape enters through the skin. While titanium dioxide itself is a valuable, non-poisonous compound, titanium waste is to a great degree acidic and its transfer techniques which lead to a lot of various ecological issues. For example in a case, Most beach front generation plants utilizing the sulfate procedure dump a lot of sulfuric corrosive into the North Sea. Although the soluble ocean water cushions and kills the weaken acidic waste, dumping sulfuric corrosive causes a sudden drop in the pH estimation of the getting water and decreases the oxygen substance of the water, consequently this process leads in destroying of marine life.
References:
Ireland, J.C. et. al. Inactivation of Escheria coli by Titanium Dioxide Photocatalytic Oxidation.
Applied and Environmental Microbiology.
- Ireland, J.C… et. al. Inactivation of Escheria coli by Titanium Dioxide Photocatalytic Oxidation.
- . Li SQ, Zhu RR, Zhu H..,, Xue M, Sun XY, et al…. Nanotoxicity of TiO2 nanoparticles to erythrocyte in vitro. Food Chem Toxicol. 2008;46:3626-3631.
- Buzea C,…. Pacheco II, Robbie K. Nanomaterials and nanoparticles: sources and toxicity. Biointerphases. 2007;2:17-71
- Zhao J, Bowman L, Zhang X, Vallyathan V, Young SH, et al….. Titanium dioxide (TiO2). J Toxicol Environ Health A.
- Panigrahi S, Basak D. Core-she,,.,ll TiO2@… ZnO nanorods for efficient ultraviolet photodetection. Nanoscale
- Yoo K-C, Yoon C-H, Kwon D, Hyun K-H, Woo,.,., SJ, et al. Titanium dioxide.
- Liao D, Badour C, Liao B.,.,. Preparation of nanosized TiO2/ZnO composite catalyst and J photochem photobiol A Chem
- www.nanopartikel.info/en/nanoinfo/materials/titanium-dioxide/overview
- copublications.greenfacts.org/en/titanium-dioxide-nanoparticles/
- Zhao.. C, Deng H, Li Y, Liu Z. Photodegradation of oxytetracycline in aqueous by 5A and 13X loaded with TiO2 under UV irradiation. ..,J Hazard Mater.
- Moon J, Yun CY, ..Chung K-W, Kang M-S, Yi J. Photocatalytic activation of TiO2 under visible light using Acid Red 44… Catalysis Today.
- Zhang J, Wages M, Cox SB, Maul JD, Li Y, et al. Effect of titanium dioxide nanomaterials.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allDMCA / Removal Request
If you are the original writer of this essay and no longer wish to have your work published on UKEssays.com then please click the following link to email our support team:
Request essay removal


