The Inhibitory Effects of Low Power Radiofrequency
| ✅ Paper Type: Free Essay | ✅ Subject: Physics |
| ✅ Wordcount: 1862 words | ✅ Published: 12 Mar 2018 |
Inhibitory effects of low thermal radiofrequency radiation on physiological parameters of Zea mays seedlings grown
Mihaela Răcuciu1, Cora Iftode2 and Simona Miclăuş3
Abstract.
The inhibitory effects of low power radiofrequency field on physiological parameters of Zea mays plantlets developed from exposed seeds were obtained in present experiment. Identical biological samples of 30 Zea mays seeds with a uniform genophond were exposed to a continuous wave at a frequency of 1GHz, inside a transverse electromagnetic (TEM) cell, for different exposure times between 0 and 8 hours, and then let to germinate. By numerical dosimetry computations, an average specific absorption rate (SAR) of energy deposition per sample of 0.47W/kg was obtained and SAR distribution over the sample was determined. The assimilatory pigments (chlorophyll a, chlorophyll b and total carotenoid pigments) and average nucleic acids (DNA and RNA) contents in the green tissues of all experimental samples as well as in the control plants were assayed by spectrophotometric methods, after 12 days of plant growth. Decreased levels of all assimilatory pigments and nucleic acids were obtained for all exposure durations, as compared to the control sample. Also, it was found that the enhanced exposure time had an inhibitory effect on the growth of the 12 day old plantlets.
- Introduction
The scientific interest about the effects of high frequency electromagnetic fields on biological materials, especially plants, dates since the 19th century [1]. Not a few of the earlier experiments on plant material have been focused on the effect of radio frequency electromagnetic fields on seeds. In many cases, the short exposure resulted in increased germination rate and stimulation of seedlings growth [2]. Ponomarev et al. [3] investigated the influence of low intensity microwave radiation on the germination of cereals (winter and spring wheat, spring barley, oats), observing an increasing of germination rate for all the treated seeds. Khalafallah et al. showed in their experiment that the germination grains and growth rate of exposed maize seedlings, significantly increased compared to the control. Also, photosynthetic pigments levels, total soluble sugar and total carbohydrates were positively affected by 945MHz electromagnetic field exposure [4]. Jonas reported that the action of microwave radiations on Zea mays seedlings damaged the photosynthetic system and led to significant increase of the carotene and anthocyanin production [5].
Tkalec et al. exposed Lemna minor L. plants to 900 MHz electromagnetic field and then was observed a decreasing of the plants growth for 2 hours exposure, even if the germination rate and the root lengths not changed significantly [6]. M. Ursache et al. [7] exposed Zea mays seedlings at 418 MHz electromagnetic field for relatively short exposure times (1 to 4 hours), and observed increasing of the photosynthetic pigments levels. L. Oprică [8] has studied microwave treatment with power density under 1 mW/cm3 on rapeseeds and concluded that the exposure determined variations of catalase and peroxidase activities depending on the age of the plants and time of exposure. Sandu et al. [9] studied the 400MHz electromagnetic field influence on the black locust (Robinia pseudoacacia) seedlings. Chlorophyll a as well as chlorophyll b level was found to decrease and chlorophyll ratio was decreasing logarithmically to the increase of daily exposure time on the electromagnetic field. Roux et al. [10] showed that a non-thermal 900MHz electromagnetic field is able to evoke rapid accumulation of some transcripts (like Ca2+ receptors) that are known to play a role in the early events of plant responses to stress in tomato plants.
The present study aimed to quantify the effects of low-power 1GHz electromagnetic field action on Zea mays seeds, by assessing of the photosynthetic pigments and nucleic acid total level in seedlings developed from exposed seeds. The seeds exposure took place in a well-controlled environment and was seconded by a proper microwave dosimetric analysis.
- Materials and methods
Biological material. Considering its economic importance for agriculture and food industry the popcorn seeds (Zea mays) were chosen as biological material. In order to diminish the putative genophond variations in this experiment were used seeds from a single plant with vigorous biological features from an experimental micropopulation. Irradiated seeds germination occurred on porous paper support, in darkness and closed Petri dishes, environmental conditions being kept under peer control (temperature being 24±0.50 C and 98% humidity). After germination the young plantlets development was conducted in the same controlled laboratory conditions (t=23±0.50C, illumination -11h: 13h light/dark cycle and 90% humidity) and the culture medium of young plantlets was daily watered with the same amount of deionized water.
Exposure system and modelling-simulation. Biological samples composed of 30 Zea mays seeds each, having a uniform genophond, were exposed one by one (in a Petri dish) to 1GHz microwave, inside a transverse electromagnetic (TEM) cell, for different exposure times between 1 hour and 8 hours (Fig. 1). The TEM cell was model IFI CC-104SEXX (Instruments for Industry, USA) and at the input port the incident power Pin=11.5W was delivered from a radiofrequency signal generator model Hameg HM 3184-3 through a power amplifier Ophir 5150. At the output port the cell was terminated by a 50 ohm matched impedance. In this way, a relatively uniform electric (E) field strength distribution was obtained in a large volume between the floor and the septum of the TEM cell [x], at the place where the sample was exposed.
A modelling-simulation step followed, in order to obtain the specific absorption rate of energy deposition (SAR) in the sample. The validation of the approach [x] showed that in general a good agreement between measured and simulated SAR is obtained. In present case, the simulation was made in CST Microwave Studio. The simulated TEM cell and the sample disposal inside it is observed in Fig. 1a, while in Fig.1b the distribution of the seeds in the Petri dish during exposure is indicated. Getting primary data from the real sample, each corn seed was then modelled as a little ellipsoid, with the axes of 1×0.8×0.41 cm, and with an average mass of 0.317g. The mass density of a corn seed was of 1.8628g/cm3. The dielectric properties of the seeds were extracted from [xx], by considering an average of 23% relative humidity of the sample. So, the dielectric data for Zea mays were: relative permittivity real part – which is related to the stored energy, was ε’r=7.15, relative permittivity – imaginary part, which is related to the dissipation (or loss) of energy was ε”r =1.52, and the electric conductivity was σ=84.5×10-3 S/m.


- (b)
Fig.1. The modeled TEM cell and the disposal of the corn seeds sample inside it, for uniform exposure to 1GHz/11.5W: a) the position of the Petri dish inside the cell during the exposure; b) the disposal of the seeds inside the dish
Analysis methods. The assimilatory pigments (chlorophyll a, chlorophyll b and total carotenoid pigments) and average nucleic acids (DNA and RNA) levels in the green tissues of all experimental samples as well as in the control plants were assayed by spectrophotometric methods using a JASCO V530 spectrophotometer UV-VIS device provided with quartz cells of 1cm width, after the 12 days of plant growth. Using the Lichtenthaler and Welburn’s method [11], the assay of the assimilatory pigments extracts (in 80% acetone) was performed, while the assay of nucleic acid level (in perchloric acid 6% extracts) was carried out accordingly to modified Spirin’s method (Spirin [12]; Struchkov et al. [13]). Spectrophotometric measurements were performed at the wavelengths of: 663nm, 646nm and 470nm (versus acetone 80%) for the assay of chlorophylls (Chla, Chlb) and carotenoids pigments (Car) from green tissues and, at 260nm and 280nm (versus perchloric acid 6%) in the case of nucleic acids. For obtaining of photosynthetic pigments levels, the formulas from Lichtenthaler and Welburn [11] were applied while the calibration curves (based on the spectral readings to the mentioned wavelengths) were used in the case of nucleic acids. The biological material, used in the analysis conducted in this study, was consisted of green tissue obtained by mixing up the green tissue from the all young plantlets grown from each experimental group (sample). It was used the same amount of fresh green tissue mass for each experimental sample. Plant individual length was measured with 0.1cm precision and the average lengths and the standard deviations were calculated for each batch of test seeds. Using the Student test, the confidence interval was calculated for every batch of plantlets for the confidence levels P = 90%, 95% and 99%. Since chlorophylls ratio revealed the response of the LHC II system (Light Harvesting Complex II) to the external stimuli, these experimental data offered the main insight into the photosynthesis complex processes (Ort et al.[14]).
Statistic analysis. Statistic analysis of the experimental data, resulted from the three repetitions of the whole experiment, was accomplished by means of ANOVA test – applied using MsExcell soft package – to evaluate reliability of modifications induced by electromagnetic field exposure in comparison to the control ones as well as among the samples corresponding to different exposure time, considering the significance criterion of 0.05 (p value).
- Results and discussions
The SAR distribution, obtained in the simulation (Fig.2), was not very uniform, varying between 0.27-0.65W/kg, with an average value SARavg=0.47W/kg. With this SAR value, and by considering a specific heat of the corn c=3350 J/kg.degC, an expected temperature increase due to microwave exposures between 0.50degC and 4.04degC is expected, when no heat exchange would take place between the seeds sample and the environment. This would conduct to a low-thermal effect.

Fig.2. SAR distribution along the 30 seeds probe exposed in the TEM cell at 1GHz
The average lengths of plantlets and afferent standard deviations were calculated for each batch of test seeds and represent in Figure 2 in function of electromagnetic field exposure time. It was found an inhibitory effect on plant growth under the 1GHz electromagnetic field action with enhanced exposure times. The confidence interval was calculated for every batch of plantlets using the Student test, for the confidence level P = 90%. All length plantlet results are statistically significant in comparison to control.

Fig.2. The average length of 12 days old plantlets in function of exposure time
The contents of photosynthesis pigments (a and b chlorophylls and total carotenoids) in the green tissue of young Zea mays plantlets (aged of 12 days) for experimental samples in Fig. 3 are presented. The chlorophyll a level, the main photosynthesis pigment, was found decreased for all electromagnetic field exposure times used in this experiment comparatively to the control sample (plants growth was performed only in deionised water presence) (statistically significant in relation to the threshold of 0.05).

Fig. 3. Assimilatory pigments level in Zea mays plantlets versus 1GHz electromagnetic field exposure time. (Chl a –the content of chlorophyll a, Chl b – the content of chlorophyll b, Car – the content of total carotenoid pigments).
The total assimilatory pigments contents had the same variation to the increase of exposure time of electromagnetic field action on 12 days young plantlets that was observed for chlorophyll a level. The chlorophylls ratio (chlorophyll a / chlorophyll b) is considered the best indicator upon the photosynthesis process efficiency (Ort et al.[14]) which provides indirect information on the enzymatic aggregates of the Light Harvesting Complex II (LHC II) from the photosynthetic system II located in the chloroplasts membranes. A slight variation of chlorophylls ratio was observed for enhanced exposure time only for the greater exposure time (8 hours) was observed decreased value, with 22% than unexposed sample value (statistical significance was ensured relatively to the threshold of p<0.05).
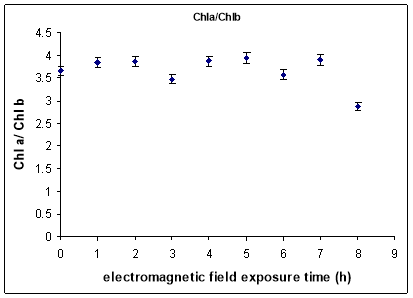
Fig. 4. The effects of electromagnetic field exposure on chlorophylls ratio (Chl a/Chl b)
From results, it was observed for relatively small exposure times a tendency to accumulation of more chlorophylls than carotenoids while for enhanced exposure times (between 4 and 8 hours) this tendency has been changed, observing an accumulation of more carotenoids than chlorophylls at plant level. Chlorophylls to carotenoids ratio decreased with 1GHz electromagnetic field exposure time (Fig. 5).
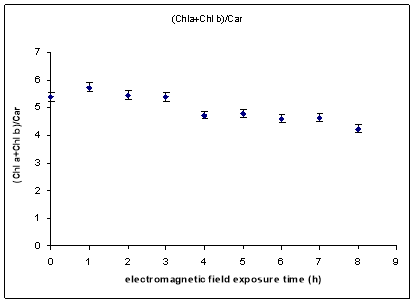
Fig.5. (Chl a+Chl b)/Car level for the plantlets provided by electromagnetic field exposed seeds
The nucleic acids average content in young Zea mays plantlets after 12 days of grown developed from 1GHz electromagnetic field exposed seeds is presented in Fig. 6. It was observed that for increasing electromagnetic field exposure time the nucleic acid biosynthesis was inhibited (about 75 %) in comparison to the control sample (plants developed from unexposed seeds). Applying the t-test to compare control and test sample, data for the average nucleic acid level statistic significance (p<0.05) was found for all samples. Nucleic acid total level decreased exponentially with exposure time t, experimental fitting curves following 20.72 + 42.37e-t/1.44 equation with R2=0.94227.
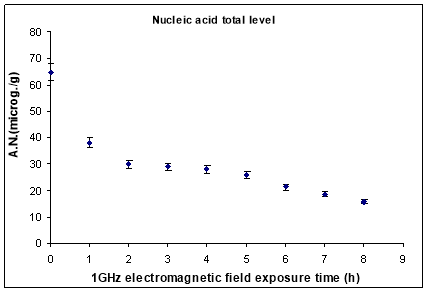
Fig. 6. The level of DNA and RNA for the plantlets provided by electromagnetic field exposed seeds
- Conclusions
References
[1] P. A. Ark, W. Parry, “Application of high-frequency electrostatic fields in agriculture,” The Quarterly Review of Biology, vol. 15, no. 2, pp. 172–191, 1940.
[2] V. N. Tran, “Effects of microwave energy on the strophiole, seed coat and germination of acacia seeds,” Australian Journal of Plant Physiology, vol. 6, no. 3, pp. 277–287, 1979.
[3] Ponomarev, L. I., V. Е. Dolgodvorov, V. V. Popov, S. V. Rodin, О. Ð. Roman, The effect of low-intensity electromagnetic microwave field on seed germination (in Russian), Proceedings of Timiryazev Agricultural Academy, 1996, 2, 42–46.
[4] A.A. Khalafallah, Samira M. Sallam, Response of maize seedlings to microwave at 945MHz, Romanian Journal of Biophysics, Vol. 19, No. 1, P. 49–62, 2009
[5] Jonas, H., Responses of maize seedlings to microwave irradiations, Environmental Pollution Series B. Chemical and Physical, 1983, 6, 207–219.
[6] M. Tkalec, K. Malaric, B. Pevalek-Koylina, Influence of 400, 900, and 1900 MHz electromagnetic fields on Lemna minor growth and peroxidase activity, Bioelectromagnetics, vol.26(3), pp.185–193, 2005.
[7] M. Ursache, G. Mindru, D.E. Creanga, F.M. Tufescu, C. Goiceanu, The effects of high frequency electromagnetic waves on the vegetal organisms, Rom. Journ. Phys., Vol. 54, Nos. 1–2, P. 133–145, 2009
[8] L., Oprica, Effect of microwave on the dynamics of some oxidoreductase enzymes in Brassica napus germination seeds, „Alexandru Ioan Cuza” University Scientific Annals genetic and Molecular Biology Section, 2008, IX(4), 11–16.
[9] D. D. Sandu, C. Goiceanu, A. Ispas, I. Creanga, S. Miclaus, D.E. Creanga, A preliminary study on ultra high frequency electromagnetic fields effect on black locust chlorophylls, Acta Biologica Hungarica, 56(1/2) (2005), 109-117.
[10] D. Roux, A.Vian, S. Girard, P. Bonnet, F. Paladian, E. Davies, G. Ledoigt, Electromagnetic fields (900MHz) evoke consistent molecular responses in tomato plants, Physiologia Plantarum, vol. 128, pp.283-288, 2006.
[11] H.K. Lichtenthaler, A.R. Wellburn, Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents, Biochem. Soc. Transact., 11, 591 – 559 (1983)
[12] A. Spirin, Spectrophotometric determination of total nucleic acids, Biokhimiya (USSR), 23, 656–662 (1958)
[13] V.A. Struchkov, N.B. Strazhevskaya, R.I. Zhdanov, DNA-bound lipids of normal and tumor cells: retrospective and outlooks for functional genomics, Bioelectrochemistry, 58, 23-30 (2002)
[14] D. Ort, J. Whitmarsh, Photosynthesis. Encyclopedia of Life Sciences. Macmillan, London; 2001.
[x] C. Iftode C., S. Miclaus, “Design and Validation of a TEM Cell Used for Radiofrequency Dosimetric Studies”, Progress in Electromagnetic Research – PIER, vol. 132, pp.369-388, 2012.
[xx] E. Surducan, C. Neamtu, V. Surducan, Dielectric properties of Zea mays kernels – studies for Microwave Power Processing Applications, Processes in Isotopes and Molecules, Journal of Physics: Conference Series 182 (2009) 012017, IOP Publishing, 2009.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allDMCA / Removal Request
If you are the original writer of this essay and no longer wish to have your work published on UKEssays.com then please click the following link to email our support team:
Request essay removal


