Pellet Catalyst Development using Ferroxcube Process
| ✅ Paper Type: Free Essay | ✅ Subject: Engineering |
| ✅ Wordcount: 186 words | ✅ Published: 31 Aug 2017 |
The pellet preparation followed the classic routine of the powder metallurgy industry (NTUA): hematite (Fe2O3) or magnetite (Fe3O4) powders were homogenized in a plasticizer and greasing environment, within a rotating pan (Figure 7). Consequently, disks were pressed in a 150 bar hydraulic press (Figure 8). NTUA used the experience of CERTH (coordinators of the HYDROSOL projects to proceed in the proper preparation of the pellets).


Figure 7. Rotating panFigure 8. The 150 bar hydraulic press
After the initial pelletization by the hydraulic press, the pellets were undergone sintering and reduction in a controlled atmosphere (oxygen-reduced atmosphere) oven as shown in Figure 9, in order to obtain the magnetite phase certified by XRD, as shown in Figure 10.

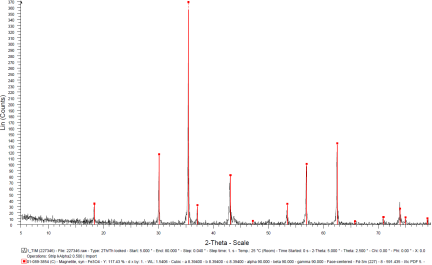
Figure 9. The sintering oven.Figure 10. XRD response of the sintered pellet, certifying the magnetite phase
The porosity of the pellet and the neck development, offering substantial mechanical properties, have been monitored by SEM, as shown in Figure 11, as well as by the classic pressurizing methods.
This way, the magnetite pellets have been stabilized in mechanical, electric and magnetic properties, offering mechanical robustness and electric resistivity below 10 Ohm or <103 ïOhm.m (non-used pellet).
Furthermore, an advanced manufacturing process has been proposed by the SUN Unit to the above-mentioned industrial technique. During sintering and reduction process, magnetic field can be added externally to the sintering and reducing ovens to magnetically orient the magnetite dipoles in the desired direction, allowing for advanced Lorentz force control.
2.2. A thin-film catalyst
We have conceived and developed a thin film device for the hydrogen production which can be integrated in an automated system. The device can also be used for the reduction and oxidation process in oxide semiconducting films supported by metal substrates-wafers, even if they do not exhibit coherent magnetic order. The method is based on the Lorentz forces acted on the reducing – oxidizing conducting electrons in the said oxide semiconducting films. Hence, this device can trap oxygen and hydroxyl from transmitted water, thus producing hydrogen. This such reduction and oxidation process in open air, as well as controlled atmosphere conditions.
A particular application, useful for the HELENIC REF project refers to the three-layer depicted in Figure 15. The upper layer consists of a magnetite film not thicker than 500 nm, an insulating thin film supporting the magnetite film, not thicker than 500 nm and finally a metallic wafer made of Al or other metal, supporting the insulating and the magnetite thin film.






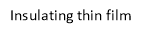


Figure 15. Schematic of the catalyst
Possible manufacturing methods include electrodeposition, physical vapor deposition, chemical vapor deposition as well as lithography and masking methods for the generation of electric contacts on the upper thin film.
Concerning Fe3O4 (magnetite) film, a preferable but not obligatory condition is to be able to provide magnetic order in all grains of the ferrimagnetic cells of the oxide. This is achieved by magnetic field during deposition or by post-processing magnetic field annealing with subsequent field cooling.
The desired properties of the Fe3O4 (magnetite) film are indicatively as follows:
- Structure of the magnetite phase, which can be certified by X-ray powder diffraction
- Multi-grained microstructure with small necks between grains, monitored by Electron Microscopy
- Resistivity below 10 kOhm, indicative of semiconducting phase
- Magnetic order, observed by monitoring the magnetic hysteresis loop
- Mechanical robustness observed by hardness and wear measurement
Applying electric current through the metallic support of the film results in magnetic field parallel to the surface of the film and vertical to the applied electric current. The amplitude of this field is:
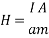
Where I is the applied electric current and a the thickness of the film. If the thickness a (sum of the magnetite film and the insulating thin film) are less than 1 ïm and the current I is C Amperes, then the applied field is:
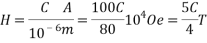
Thus, the applied field is controllable by the applied electric current through the metallic support. This current can easily be in the order of one Amperes in dc mode or several tens of Amperes in pulsed mode, if the thickness of the metallic support is in the order of 100 ïm, thus allowing the transmission of magnetic field larger than 1 T in the vicinity of the oxide semiconducting film.
Therefore, passing electric current through metallic support, one can simulate the magnetic field vertical to the direction of electric current passing through the magnetite thin film. The directions of these two electric currents characterizes the direction of the Lorentz force on the conducting electrons of the magnetite film. Parallel currents result in forcing the electrons towards the insulating thin film, while anti-parallel currents force them towards the surface of the magnetite film. These two different directions of Lorentz forces have a different effect on the operation of the magnetite thin film, acting as catalyst for the production of hydrogen.
Passing electric current along the magnetite film, independent on the electric current passing through the metallic substrate of the magnetite film, (i.e. from another source), along the same direction of the current transmitted to the metallic substrate, results in a Lorentz force acting on the charge carriers (electrons) moving them from the surface towards the insulating thin film. Thus, the surface of the magnetite film becomes activated, in other words is reduced due to the removal of the mentioned Lorentz electrons.
Activation is the generation of electron vacancies, offering the generation of new oxygen vacancies (excess of oxygen vacancies). Thus, the resistivity drops, and the applied voltage results in higher current, which increases the local temperature to 280 – 320C. Such temperature increase is observable by any kind of pyrometer. The temperature increase is dependent on the combination of the electric current passing through the oxide semiconducting film and the magnetic field transmitted to it due to the electric current passing through the metallic support.
The effect is reversible and un-hysteretic: increase or decrease of the amplitude of I x H product, where I is the applied current on the oxide film and H the field resulting from the current on the metallic substrate, results in un-hysteretic and reversible increase or decrease of the temperature and reduction of the oxide film respectively.
Such activation can be maintained even in open air: the competition between the Lorentz electrons based reduction and the free air oxidation finally results in oxidation which can also be macroscopically observed by the decrease of the activated (reduced) area on the free surface of the film. Having limited the current passing through the metallic support may result in activation (reduction) shrinkage. In this case, increasing the current transmitted to the metallic substrate before the radiation disappears, or before oxygen vacancy excess disappears, the oxygen vacancy excess is rejuvenated. Such rejuvenation is completely reversible by using the magnetic field produced by the current passing through the metallic support (in the order of 1 T).
After complete oxidation (phase transformation of all the Fe3O4 film to Fe2O3) such oxygen vacancy excess and consequent reduction of the compound is impossible with magnetic fields of this order.
The reduction and oxidation process can be monitored by measuring the surface resistivity of the magnetite film. Such resistivity depicts the increase or decrease of oxygen vacancies on the surface and consequently the bulk of the oxide pellet. Increase and decrease of this resistivity is equivalent to the oxidation and reduction of the resistivity of the surface respectively.
This effect is observable in all types of oxide semiconducting films even in the absence of coherent magnetic order in them. To be able to observe this effect in oxide semiconducting films, they should be in a continuous solid state form, exhibiting even a small electric conductivity, and provided that magnetic field and electric current of proper waveform and amplitude are simultaneously applied on the semiconducting oxide film and the conducting (metallic) support.
Such reduction mechanism can be used in the reduction of compounds passing on or through the film surface. Our focus is the reduction of pure vapor water to H2, by means of trapping the oxygen or the hydroxyl of the water molecule to the reduced (or activated) magnetite. Such oxidation with consequent hydrogen production can be reversed back to oxide reduction (activation) by means of stopping the water vapor transmission. This way, the two electric currents will reduce (activate) again the surface of the oxide film, provided that no water vapors are transmitted to the pellet surface or body.
A certain application of such effect is the use of the said pair of Fe3O4 (magnetite) film with its metallic support for the production of hydrogen and oxygen from water vapors, using the following algorithm:
- Independent electric currents pass through the metallic support and the oxide semiconducting film in the same direction. Thus, the surface of the oxide semiconducting film is reduced (activated).
- Then, water vapor is transmitted on the surface of the oxide semiconducting film, resulting in oxygen trapping, hydrogen production and oxidation of the surface of the oxide semiconducting film.
- As soon as the resistance of the surface of the oxide semiconducting film exceeds a certain threshold, indicating reach to a critical level of oxide film surface reduction, the water vapor transmission is stopped.
- Thus, the oxide film surface is re-activated, followed by consequent oxygen release, until the resistance of the oxide film surface reaches a minimum threshold, indicating proper reduction (activation), which is proper for oxygen trapping.
- Then, water vapor is re-transmitted to the re-activated oxide film surface for hydrogen production.
The production of hydrogen and oxygen is followed by an automatic storage of these two gases in different chambers to be used for any scope, like thermal combustion or fuel cell operation or other purpose.
Development of the thin film catalysts
The Dynamic Hydrogen Bubble Template (DHBT) is an excellent method for the fabrication of macrostructures with nanoporous side walls. The method has been named from the phenomena when a series of hydrogen-bubbles generated on the surface of the substrae act like a dynamic negative template. Figure 1 demonstrates the theoretical approach of the technique. Figure 1(a) illustrates the hydrogen-evolution taking place on the surface there is no metal/metal-oxide deposition. The size of the bubbles increasing along the distance from the substrate surface. Fig 1(b) shows the SEM-image of copper nanofoams prepared by DHBT.
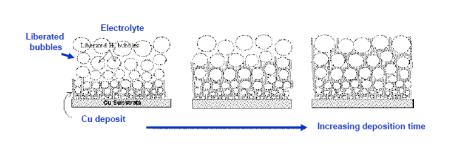

Figure 1. (a) Simplified description of DHBT generated metal/metal-based foams; (b) Honeycomb-like Cu-deposit structure made by DHBT.
The electrochemical deposition procedure of the development of the catalyst is divided in two main parts. At first preliminary experiments on deposition of magnetite film were carried out in order to determine the orders of amplitude of the different involved parameters (current density, geometry etc.). , the electrochemical deposition of iron-oxide films was carried out as a preliminary approach. Based on the results of the characterization, the parameters of the deposition were varied in a high range in order to obtain 3D-structured porous nanofoams.
The films were electrodeposited onto copper substrates (purity: 99.5%). The substrates were mechanically polished with 800 and 2000 emery papers, respectively. Henceforward, the polished substrates were ultrasonically cleaned in deionized water, ethanol and acetone, respectively. The 2 cm2 active surface area of the substrate was ensured by commercial nail-polish and insulating tape. The galvanostatic deposition was carried out in a two-electrode cell using a DC Power Supply. Platinum-mesh was used as counter electrode.
Two different arrangement of the working electrode have been tested as shown in Figure 2. The electrolytic bath was a strongly alkaline solution with pH 12.5 made of 0.037 M Fe2(SO4)3.x H2O, 0.1 M triethanolamine (TEA) and 2 M NaOH. After mixing the components at T = 80 ï‚°C the solution had greyish-green color and no turbidity was observed.
The parameters of the deposition process were varied in the terms of current density, bath temperature and deposition time. The range of cathodic current density was chosen j = 5-15 mA.cm-2, the depostion time was given from 5 min to 6 hours and the bath temperature was adjusted T = 60-90 ï‚°C. The nominal thicknesses of the prepared structures were calculated by using the Faraday-law.
The surface morphology of the deposited film-layers were studied by scanning electron microscope (Nova NanoSEM 230) and the crystallographic characterization was determined by X-ray diffraction (Bruker D8 Advance, Cu radiation-40kV, 35mA).
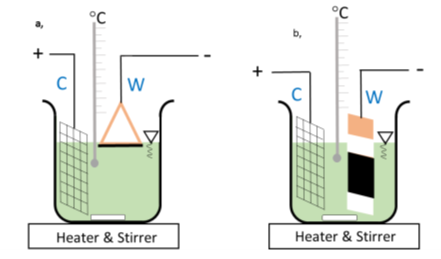
Figure 16. a) copper triangle used as working electrode (W) by turning the active surface towards the surface of the electrolyte b) copper-plate (W) used as working electrode by immersing it totally and by turning the active surface towards the counter electrode (C).
Concerning the development of 3D-structured porous films, the substrate deposition was not changed (Cu-foil, purity: 99.5%). SiC polishing papers were used in grade of 800, 1500, 2500 and 4000, respectively as a surface pre-treatment. All the pre-treated samples were ultrasonically cleaned in deionized water, ethanol and acetone, respectively. In that case, the active surface area of the substrates was reduced to 0.5 cm-2 and fixed with nail-polish and insulating tape. The galvanostatic deposition was carried out in a two electrode cell using a BioLogic SP 300 potentiostat. Platinum-wire was used as counter electrode.
Concerning the result of the film-deposition the arrangement of the working electrode was set in vertical position. The electrolytic bath, concentrations and its properties were the same as described above. The cathodic current density was varied in the range ofj = 0.075-1 A.cm-2, the depostion time was changed between 30 sec to 180 s and the bath temperature was adjusted T = 90 ï‚°C. Similar films have also been developed by using sputtering deposition and electron-gun aided deposition (Figure 17).
Apart from the electrolytic method, thin magnetite films were also deposited onto insulating films by usign spuutering and electron gun physical evaporation at the NTUA. However, the obtained results on the porosity of the magnetite films are not comparable with the DHBT method and therefore they are not demonstratable at their current stage. Nevertheless, experiments are under way to obtain PVD films of the quality observed in the electrodeposited films.
- Characterization of catalysts
- Structural analysis
The catalyst pellets were prepared by a proprietary process from Fe3O4 powders. IP SAS used Fe3O4 powder with declared purity better than 99% and nominal grain size of 2-4um supplied by chemicalstore.com, product code FE3O4M1. The phase composition determined by XRD (X-Ray Diffraction) is shown in Figure 38. Minor traces of Hematite can be observed at 32.8° 2Theta for the sample produced at IP SAS.
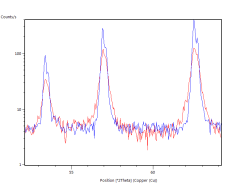
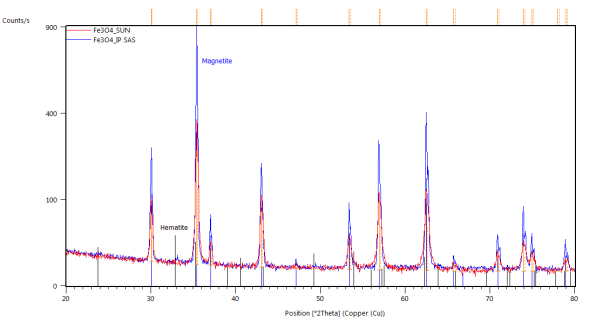
Figure 38. XRD patterns from pellets prepared by SUN (red) and IP SAS (blue). Marked constituent phases are Magnetite (blue lines) and Hematite (black lines)
A typical XRD response in the bulk SUN pellets and in bulk CERTH pellets is illustrated in Figure 1, demonstrating exclusive magnetite presence.
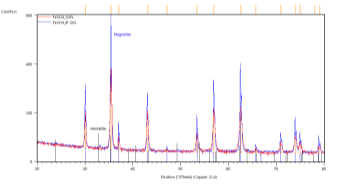
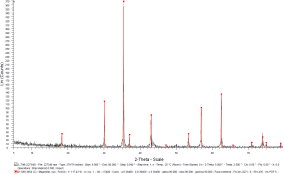
Figure 1. XRD response of bulk pellets from SUN (left) and CERTH (right) illustrating exclusive presence of magnetite
Phase transformations were studied by the means of in-situ XRD presented in Figure 41. Samples were mortar milled to obtain a fine powder that could be spread on the sample holder of the furnace. Milling force was kept minimal to preserve the structure of the pellet parts. Linear heating with indicated rate was used to obtain well-defined diffraction maxima with respect to the signal to noise ratio. Diffractometer was configured for parallel beam illumination with a secondary LiF monochromator to supress fluorescence from the Fe containing pellets.
Formerly purely magnetite pellets are transforming to hematite in the vicinity of 300°C. After the transformation no other phase change was observed. Pellets supplied by SUN exibit the transformation at higher temparature than the pellets prepared at IP SAS. This can be atributed to the smaller size of the Fe3O4 crystallites in the SUN pellets.
Detailed pattern examination is shown in Figure 42. The studied powdered pellets exhibit only presence of magnetite and hematite respectively. Diffraction maxima at 26° 2Theta corresponds to SiO2 present as a thin layer grown on the non-reflective Si sample holder.
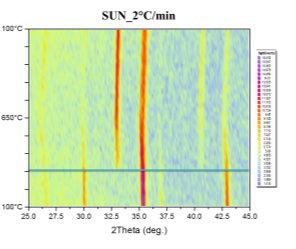
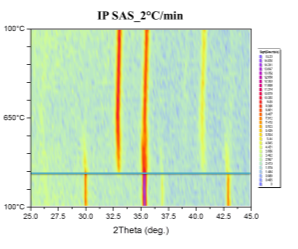
Figure 41. In-situ XRD patterns of catalysts in air atmosphere with indicated heating rate
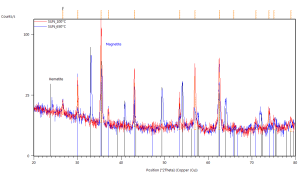
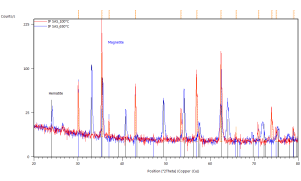
Figure 42. In-situ XRD patterns of annealed catalyst pellets at indicated temperatures.
Marked constituent phases Magnetite (blue lines) and Hematite (black lines).
Catalyst prepared at SUN left, IP SAS right.
Crystallographic study of the iron-oxide thin films samples was also realized. Figure 4 represents the identification of the observed peaks. Figure 4(a) shows the results of sample prepared in 3300 s in 70 ï‚°C bath with -5 mA.cm-2 current density. Grazing incidence XRD patterns of this sample indicate the presence of magnetite (blue lines) and copper substrate (green lines). Grazing angle was 2 (red curve) and 6 degrees (blue curve), respectively.
In Figure 4(b) the results of sample with same conditions as in Figure 4(a) can be seen, but in this case the current density was -9 mA.cm-2. Here, grazing incidence XRD patterns indicate iron (grey lines) and copper (green lines). Grazing angle was 2 (red curve) and 6 degrees (blue curve), respectively. Figure 4(c/1) and (c/2) show the results of XRD taken from the same sample with deposition conditions of 2 hours deposition time in 85 ï‚°C bath and -12.5 mA.cm-2 current density.
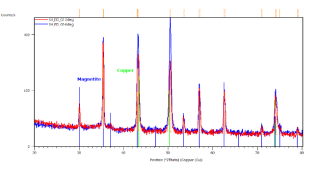
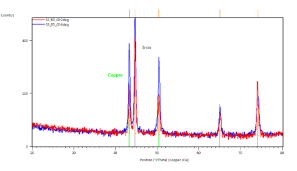
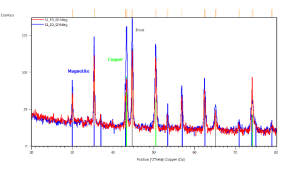
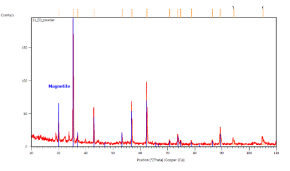
Figure 4. X-ray diffractograms of the iron-oxide electrodeposited samples prepared under different conditions. a, Δt=3300 s,T = 70 °C, j= -5 mA cm-2; b, Δt=3300 s, T= 70°C, j= -9 mA cm-2; c/1, Δt=7200 s, T= 85°C, j = -12.5 mA cm-2; c/2, powder scratched from the surface of sample c/1
In Figure 4(c/1) the grazing incidence XRD patterns were done on the sample with the substrate. Clear evidence of magnetite (blue lines), iron (grey lines) and copper as substrate (green lines). Grazing angle was 2 (red curve) and 6 degrees (blue curve), respectively. Figure 4(c/2) demonstrates grazing incidence XRD pattern of loose powder from the same sample as (c/1). Only magnetite phase (blue lines) is detected. Grazing angle was 2 degrees. The XRD results show clear evidence of magnetite if the current density does not exceed 8 mA.cm-2 cathodic current density (see Figures 4(a) and (b).
Apart from that, XPS studies have also been performed after the check meeting. For integration reasons, they are demonstrated only in the Milestone 1 document.
- Microstructural analysis
Morphology of pellets after preparation was studied by the means of SEM (scanning electron microscopy) shown in Fig. 39. The particle size is larger for the catalyst prepared by SUN. The particle size of sample prepared at IP SAS is in good agreement with the manufacturer specification and is rather homogenous in size distribution. Sample prepared at SUN is exhibiting a different size distribution ranging from very fine sub-micron particles up to particles with size increasing 50um shown in Fig. 39 and 40. As can be seen the porosity of the prepared pellets is higher for the pellet prepared at IP SAS.
The grid shown in Fig. 40 is protecting the sample from being destroyed by the magnetic field in the microscope and thus contaminating the microscope system by fine magnetic dust in the pole-piece, lens and detector system. It is worth to mention that the particle size is not directly explaining the diffraction maxima broadening shown in inset in Fig. 38. This is due to the fact that the diffraction domain size is rather different than the grain size of a particle. We can surely state that the diffraction domain size is smaller in the pellet prepared at SUN rather than at IP SAS despite the observed grain-size distribution.


Figure 39. SEM micrographs of catalysts prepared by SUN (left) and IP SAS (right)
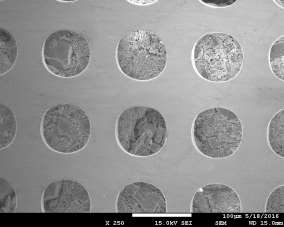

Figure 40. Low magnification micrographs of catalyst prepared by SUN (left) and IP SAS (right)
Micrographs from the CERTH pellets (Figure 41) illustrate a clear evidence of necks, responsible for the significant reduction of temperature.


Figure 41. Micrographs of the CERTH pellets, illustrating the presence of micro-necks, responsible for the improvement of the electric and mechanical properties
Concerning the thin film catalysts, SEM analysis was performed. Figure 3 demonstrates 4 different SEM-images of the prepared samples. Fig 3(a) was taken on a sample prepared in 300 s deposition period in 60°C bath with -7.5 mA cm-2 current density, and the nominal thickness was 2.24 μm. In Fig 3(b) can be seen the morphology of the sample prepared in 300 s at 70°C with-5 mA cm-2 current densitywith nominal thickness of 23.55 μm. Fig 3(c) shows the SEM-result of the sample deposited with the same bath temperature and current density as 3(b) but extended deposition time: Δt = 900 s (dn = 30.28 μm). Fig 3(d) represents the SEM-image of the sample prepared in the highest bath temperature 90°C with 5 min deposition time, -7.5 mA cm-2 current density (dn = 4.42 μm).
The SEM-images clearly show the structural changes by increasing the bath-temperature. Filamentous deposit structure has been observed at 90 ï‚°C. Due to the galvanostatic deposition mode the potential values have been not recorded. For better understanding of the structural formations potentiostatic deposition mode should be done.



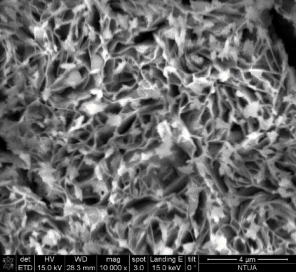

Figure 3. SEM-images of the electrodeposited iron-oxide samples with different deposition parameters (a) Δt=300 s,T= 60°C, j= -7.5 mA cm-2 at 30000X magnification (b) Δt=300 s, T= 70°C, j = -5 mA cm-2 at 15 000X magnification(c) Δt=900 s, T= 70°C, j= -5 mA cm-2at 10000X magnification (d) Δt=300 s, T= 90°C, j= -7.5 mA cm-2 at 10000X magnification.
Concerning nano-foam, due to the fact that the experiments were done in the close past the characterization of the prepared nanostructures was done only by scanning electron microscope. Figure 5 demonstrates the first promising results of the electrochemical deposition of iron-based 3D structures by using hydrogen generation on the surface as a negative template.
Such a structure was a result of applying -0.375 A.cm-2 and more negative current density, namely -1 A.cm-2 with different depositing times. Fig 5(a) to Fig 5 (c) represents a possible building process of the 3D networks with pore size range in 2-6 μm. Other type of deposit can be observed at Fig 5(d) to Fig 5(e) with numerous dendrites along the pore-sides by using current density -0.375 A.cm-2 with different deposition times. The formation of porous layer is not so obvious like in the previous case, but the size of pores are close in the same range than the pores formed at -1 A.cm-2.


Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allDMCA / Removal Request
If you are the original writer of this essay and no longer wish to have your work published on UKEssays.com then please click the following link to email our support team:
Request essay removal


